Characterization of Oils from Sandbox, Melon and Watermelon Seeds
- Popoola, C.A
- Danbaki Joshua
- 717-734
- Jun 11, 2024
- Agriculture
Characterization of Oils from Sandbox, Melon and Watermelon Seeds
Popoola, C.A*1 and Danbaki Joshua2
1Department of Chemical Engineering, Federal University Wukari, Nigeria
2Department of Food Science and Technology, Federal University Wukari, Nigeria
*Corresponding Author
DOI: https://doi.org/10.51244/IJRSI.2024.1105045
Received: 31 December 2023; Accepted: 27 January 2024; Published: 11 June 2024
ABSTRACT
This study examined the physicochemical properties and phytochemical composition of oils from sandbox (Hura crepitans), melon (Citrullus colocynthis L) and watermelon (Citrullus lanatus) seeds. Physicochemical and phytochemical analyses of the oils were carried out using American Society for Testing and Materials (ASTM) and Association of Official Analytical Chemist (AOAC) standards procedures. Sandbox oil had highest saponification value, iodine value, acid value, ester value, viscosity, cloud point and pour point with lowest moisture content, free fatty acid, peroxide value, flash point and fire point when compare with oils from melon and water melon seeds. Oils from melon and water melon seeds have low acid value, which means it can be used as a good source of edible oil. The phytochemical screening of the oils revealed the presence of saponins, deoxysugar, terpenoids, cardinolides and steroids. These results revealed that oils from sandbox seed, melon and water melon seed have great potentials as industrially promising oils, with their high saponification and iodine values, they will be good raw materials for bio-lubricant production and for a variety of industrial applications.
Keywords: Physicochemical, phytochemical, sandbox, melon, watermelon, oils.
INTRODUCTION
Vegetable oils are a group of fats that are derived from some seeds, nuts, cereals, grains and fruits. They are hydrophobic substances obtained from oilseeds, which consist mainly of triglycerides with small amounts of mono- and di-glycerides and free fatty acids, phospholids, sterols and water. They are liquids at room temperature, highly viscous, with low volatility, polyunsaturated character and suffer incomplete combustion (Aremu et al., 2015). The demand for vegetable oils is increasing at a rapid pace due to increasing demand for non-food uses of vegetable oils in biodiesels, oleochemicals, bio-lubricants and cosmetics (Baboli and Kordi, 2010). The increasing demand for oilseed products and by products has led to most oil seeds cultivation primarily for their oil and meal.
Vegetable oils are essential to meeting global nutritional demands and are utilized for many food and mineral industrial purposes. About 80 percent of the world’s vegetable oil productions are being consumed by human while the remaining 20 percent is shared between animal and chemical industries (Aremu et al., 2015). The oil from most oilseeds can be used for edible purposes like cooking and industrial production of food emulsions such as mayonnaise, margarine and creams as well as for non-edible purposes like bio-lubricants, chemicals, and pharmaceuticals. They have a rich history of use by local people as sources of food, energy, medicine and for cosmetic applications. Vegetable oils had been reported to have made an important contribution to the diet serving as a good source of protein, lipid and fatty acids for human nutrition including the repair of worn out tissues, new cells formation and as a useful source of energy (Aremu et al., 2015).
The continued increase in human population has resulted in the rise in the demand as well as the price of edible oils, leading to the search for alternative unconventional sources of oils, particularly in the developing countries. There are hundreds of underexplored plant seeds rich in oil suitable for edible or industrial purposes (Latha, 2012). The ability of a particular oilseed to fit into the growing industries depends on its utilization potential, rate of production and availability of the processing technology. The characteristics of oils from different sources depend mainly on their compositions and no oil from a single source can be suitable for all purposes (Mohammed et al., 2003).
Vegetable oils are viable bio-resource and promising candidates for the development of bio-based lubricants because they have some basic physicochemical properties which make them suitable to be used as lubricants. The use of vegetable oil as lubricant was experimented over a century ago, using oil from the agricultural industry (Oniya et al., 2014). Various studies have been carried out using different vegetable oils as raw materials for lubricant production. Naik and Galhe (2016) confirmed that the presence of monounsaturated fatty acids in vegetable oils, such as oleic and palmitic, makes it to be considered as the best feedstock for lubricants. These oils contain three hydroxyl groups and long chain unsaturated free fatty acids attached at the hydroxyl group by ester linkages that assist in dissolving more of a substance (Amdebrhan et al., 2015). The unsaturated free fatty acid which is the position and ratio of carbon to carbon double bond, three, two and one double bonds of carbon chain is named as a linolenic, linoleic and oleic fatty acid components respectively. Abere (2017) reported that vegetable oils can be used as an alternative to mineral oil or petroleum based lubricant as they provide important environmental benefits with excellent performance in many applications and possess several advantages which include biodegradability, lower toxicity, lower volatility, higher lubricity, higher flash point and viscosity index.
Phytochemicals, the biologically active and naturally-occurring compounds in plants provide health benefits for humans. They contribute to plants colour and flavor, and are found in different parts of the plants such as root, stem, leaves, fruits and seeds. They have biological properties such as antioxidant activity, antimicrobial effect, modulation of detoxifying enzymes, stimulation of the immune system, and general modulation of hormonal activity. Diets rich in phytochemicals such as carotenoids and phenolic compounds have been associated with a reduced risk of certain types of cancer, cataracts, inflammation, cardiovascular, muscular degeneration and neurodegenerative diseases (Bueno et al., 2012; Sergent et al., 2010; Snyder et al., 2011; Tanaka et al., 2012).
Sandbox tree, also known as “Dynamite tree” due to the explosive noise of the ripe fruits when split into segments, has been described as an evergreen perennial and underutilized plant which is planted as a shade tree. It is recognized by the many dark and pointed spines, smooth brown dark and spreading branches (Oniya et al., 2017). Sandbox seed according to Idowu et al. (2012) oil and contains some essential vitamins. Adewuyi et al. (2014) reported that the oil yield from the sandbox seed when compared with the yield of other oilseeds is relatively high. Okolie et al. (2012) reported the oil content of sandbox seed to be 53.61 percent (w/w), which is significant and compared favourably with other oilseeds. This relatively high oil content of the seed makes the seed a good source of oil, making it economically viable, though the seed is discarded as waste with no market value at present. This also makes it suitable for industrial applications, as any oilseeds that can produce oil up to 30 percent are regarded as suitable.
Sandbox seed oil is highly neglected non-edible oil available at little cost of production. Giwa et al. (2016) reported that it has no specific use and no commercial value presently, as the seeds are discarded as waste. The oil is a promising alternative oil because it is environmentally friendly, renewable, cheap, and easily manageable (Adewuyi, 2014). It contains high content of unsaturated fatty acid such as oleic acid which has been proved as the most ideal monounsaturated fatty acid for bio-lubricant. This makes the oil suitable for bio-lubricant production and industrial purposes because of presence of double bond in its structure will lower the melting point, which would enhance the low temperature performance of the oil.
Melon belongs to the species of the genus Citrullus of cucurbitaceae family, which usually consists of a large number of varieties (Mabaleha et al., 2007). It contains high lipid levels with 53 percent oil and 28 percent protein (Ntui et al., 2009). Various studies have reported predominantly high linoleic fatty acid content in melon seed oils. Due to the unsaturated fatty acid composition of its oil, it was reported to resemble that of corn, cottonseed, sunflower, soybean and sesame oil (Oluba et al., 2008).
Watermelon is a member of the family of cucumber (Cucurbitacea) (Oluba et al., 2008). Cucurbitaceae are known for their high protein and oil content. Watermelon seeds are sources of oils and protein with about 50 percent oil and up to 35 percent protein (Achu et al., 2005). The seeds have a high nutritive value and are a potential source of unsaturated fat, vitamins, antioxidants, minerals and proteins. The seeds have also the ability to store well, with both oil and fatty acid content being found to be stable after six months in storage (Jarret and Levy, 2012). The seeds according to Baboli and Kordi (2010) can be utilized successfully as a source of edible oils for human consumption and the oil might be an acceptable substitute for highly unsaturated oils. The seeds contain lipids of nutritional interest, with high concentrations of unsaturated fatty acids, including a high concentration of phytosterols, particularly stigmasterol and β-sitosterol (De Conto et al., 2011). The seeds also contain many beneficial minerals such as magnesium, calcium, potassium, iron, phosphorus and zinc (Yadav et al., 2011).
Selection of vegetable oils for industrial purposes relies upon the oils having relatively low cost, acceptable low temperature properties, good miscibility and acceptable oxidative and thermal stabilities. Hence, there is need for investigation on the possibility of obtaining economically viable oils from sandbox seed, melon and watermelon seed for industrial purposes. This study therefore investigated the chemical composition and phytochemical properties of oils from these three oilseeds in order to understand their potential and acceptability in industries.
MATERIALS AND METHODS
Materials
Sandbox seed (Hura crepitans) oil, melon oil and watermelon oil used for this work were collected from Wukari in Taraba State. All the solvents and reagents used for the analyses were of analytical grade and were obtained from the Food Chemistry Laboratory of the Federal University, Wukari.
Methodology for determination of the physicochemical properties
The method of Okolie et al. (2012) was adopted. The moisture content was obtained by using a known weight of the oil sample put into a clean previously weighed beaker and dried in an oven at 105°C for 4 hours. The sample was taken from the oven, cooled in a dessicator for about 30 minutes and then weighed. The procedure was repeated until a constant weight is obtained. The percentage moisture in the seed was then calculated.
Density and specific gravity
Oil of 5 ml was poured into a weighed beaker and weighed. The density was determined from the sample weight by taking the ratio of the weight of the oil to the known volume (5 ml) of the oil. Using the equation below:
Density = (sample mass)/(sample volume) (1)
Specific density = (weight of a unit volume of the oil)/(weight of equal volume of water) (2)
Refractive index of the oil was determined using ASTM D1218 method. Abbes’ refractometer was used for the measurement of the refractive index of the oil. The oil was smear on the lower position of the refractometer, after some adjustment the refractive index of the oil was read directly at room temperature (28oC).
The viscosity was measured in accordance with ASTM D445-15a (2015). This method covers the determination of viscosity using Smart series rotational viscometer TSML 21105. The viscosity was measured at room temperature (28oC). At a start a proper viscometer spindle (3) was chosen. The samples were transferred to a beaker large enough to hold the viscometer spindle. The beaker was placed on a heating mantle set to a desired temperature, while the temperature of the samples was raised. The viscosity was read at the desired temperature. The viscosities of the oils were determined using equation (3) reported by Mohammed (2015):
Viscosity=reading obtained x (factor for the spindle)/speed (3)
Saponification value
Saponification value was determined by weighing 1 gram of the oil into a flask and adding 25 cm3 of 0.1 M alcoholic potassium hydroxide solution into the flask. A reflux condenser was attached and the flask was heated on a water bath for 1 hour with constant shaking. At the end of 1 hour the flask was removed from the water bath and 1cm3 of the 1 percent phenolphthalein indicator was added. It was then titrated with the standard 0.5 M hydrochloric acid.
Saponification value = ((V₂-V₁)cm^3 x 26.05)/(weight of oil) (mgKOH/g) (4)
Acid value
Acid value of the oil was determined by mixing 25 cm3 diethyl-ether with 25 cm3 ethanol in a conical flask and 1 cm3 of 1 percent phenolphthalein indicator solution was added. The mixture was neutralized with 0.1 M potassium hydroxide solution then 1 gram of the oil was added to the neutralized solvent mixture. This was then titrated with 0.1 M potassium hydroxide solution. It was then shaken constantly until a pink colour which persists for 115 seconds was obtained.
Acid value = ((V₂ – V₁)cm³ x 5.61)/(weight of sample used) (mgKOH/g) (5)
Where: V₁ = sample titre value, V₂ = blank titre value
Ester value of oil is the number of milligrams required to saponify the ester contained in 1 gram of oil. The value was obtained as the difference between the saponification and the acid value.
Free fatty acids (FFA)
Free fatty acids were determined by weighing 1 gram of the oil samples into a conical flask. This was followed by the adding 10 cm3 of neutralized 95 percent ethanol and Phenolphthalein. This was then titrated with 0.1 M NaOH, with constant shaking until a pink colour persisted for 30 seconds. The percentage free fatty acid was calculated from the equation (6):
Free fatty acid (FFA) = (V x M x 2.82mg)/(sample weight(g)) (6)
Where: V = Volume of NaOH, M = Molarity of NaOH
2.82 = conversion factor of oleic acid.
Peroxide value
Peroxide value was determined by weighing 1 gram of the oil into a clean dry boiling tube, 1 gram of powdered potassium iodide and adding 10cm3 of the solvent mixture. The mixture was allowed to boil vigorously for 30 seconds. The tube was washed twice with 25 cm3 portions of water and the washings were added to the titration flask. This was then titrated with 0.002 M Sodium thiosulphate using starch indicator. The relation for peroxide value is given as:
Peroxide value = ((V₂-V₁)cm3 x Molarityof titrant)/(weight of oil) x 100 (meqKOH/g) (7)
Where: V₁= sample titre value, V₂ = blank titre value
Iodine value was determined by weighing 0.2 gram of the oil into a 250 cm3 glass stopper flat, and adding 10 cm3 of carbon tetrachloride to the oil and dissolved. 20 cm3 Wijs’ solution was equally added to the mixture and the content was corked with a stopper that initially moistened with potassium iodide solution. The mixture was titrated with 0.1 M standard sodium thiosulphate solution using starch as an indicator just before the end point.
Iodine value = ((V₂-V₁)cm^3 x 1.269)/(weight of oil(g)) gI₂/100g (8)
Pour point and cloud point were determined using ASTM D97 method. The oil was poured into a test tube and placed into a refrigerator to solidify. The oil was removed after it solidifies and thermometer was used to read the temperature to which the solidified oil begins to melt and flow. The lowest temperature to which the movement is observed is the pour point. The cloud point is the temperature at which a cloud of wax crystals first appear when the oil was cooled.
Flash point and fire point
The flash point measurement was done according to ASTM D93. The oil was poured into a metal container and heated at 5oC interval with a flame being passed over the surface of the sample. The temperature at which an instantaneous flash occur was taken immediately and recorded as a flash point. The fire point is that temperature at which the vapour of the oil burns constantly for 5 seconds when flame is brought near.
Methodology for determination of the phytochemical composition
The phytochemical screening of the oil samples were carried out as described by Nweze et al., (2004). The samples were screened for steroids, saponin, terpenoids, cardenolides and deoxysugar.
Equal volume of chloroform and 3 drops of concentrated sulphuric acid were added to 1 ml of oil samples. Formation of brown ring indicates the presence of steroids and formation of bluish green colour indicates the presence of phytosterols.
Saponin
Distilled water of 5 – 10 ml was added to 1 ml of oil sample and shaken in a graduated cylinder for 15 minutes lengthwise. Formation of 1 cm layer of foam indicates the presence of saponin.
The presence of terpenoids was determined by carefully adding 2 ml of concentrated H2SO4 to 1 ml of oil samples mixed with 2 ml of chloroform to form a layer. An appearance of a reddish brown colour interface indicates the presence of terpenoids.
Cardinolides
The presence of cardinolides was confirmed by adding 2 ml of glacial acetic acid containing one drop of ferric chloride solution and 1 ml of conc. sulphuric acid to the oil samples. A brown ring appearance at the interface indicate the presence of cardinolides, a violet ring appearance below the brown ring and a greenish ring in the acetic acid layer confirmed the results.
Deoxysugars
The presence of deoxysugar in the oil was determined by adding 1 ml of glacial acetic acid and 5 % ferric chloride to 2 ml of oil samples with 3 drops of concentrated sulphuric acid. Appearance of greenish blue colour indicates the presence of deoxysugars.
RESULTS AND DISCUSSION
The physicochemical properties of oils from sandbox, melon and watermelon seeds are shown in Table 1 and Figs. 1 – 7.
Table 1: Physicochemical properties of oils from sandbox seed, melon and watermelon seed
| Properties | Sandbox oil | Melon oil | Watermelon oil |
| Moisture content (%) | 0.40a | 0.40a | 0.40a |
| Density (g/cm³) | 0.90a | 0.86b | 0.82c |
| Specific gravity | 0.93a | 0.91b | 0.85c |
| Refractive index | 1.468a | 1.470a | 1.465a |
| Viscosity at 28°C (cSt) | 59.00a | 46.90b | 44.10c |
| Saponification value (mgKOH/g) | 245.00a | 170.81b | 154.98c |
| Acid value (mgKOH/g) | 2.81a | 1.99b | 1.79c |
| Ester value (mgKOH/g) | 242.19a | 168.82b | 153.19c |
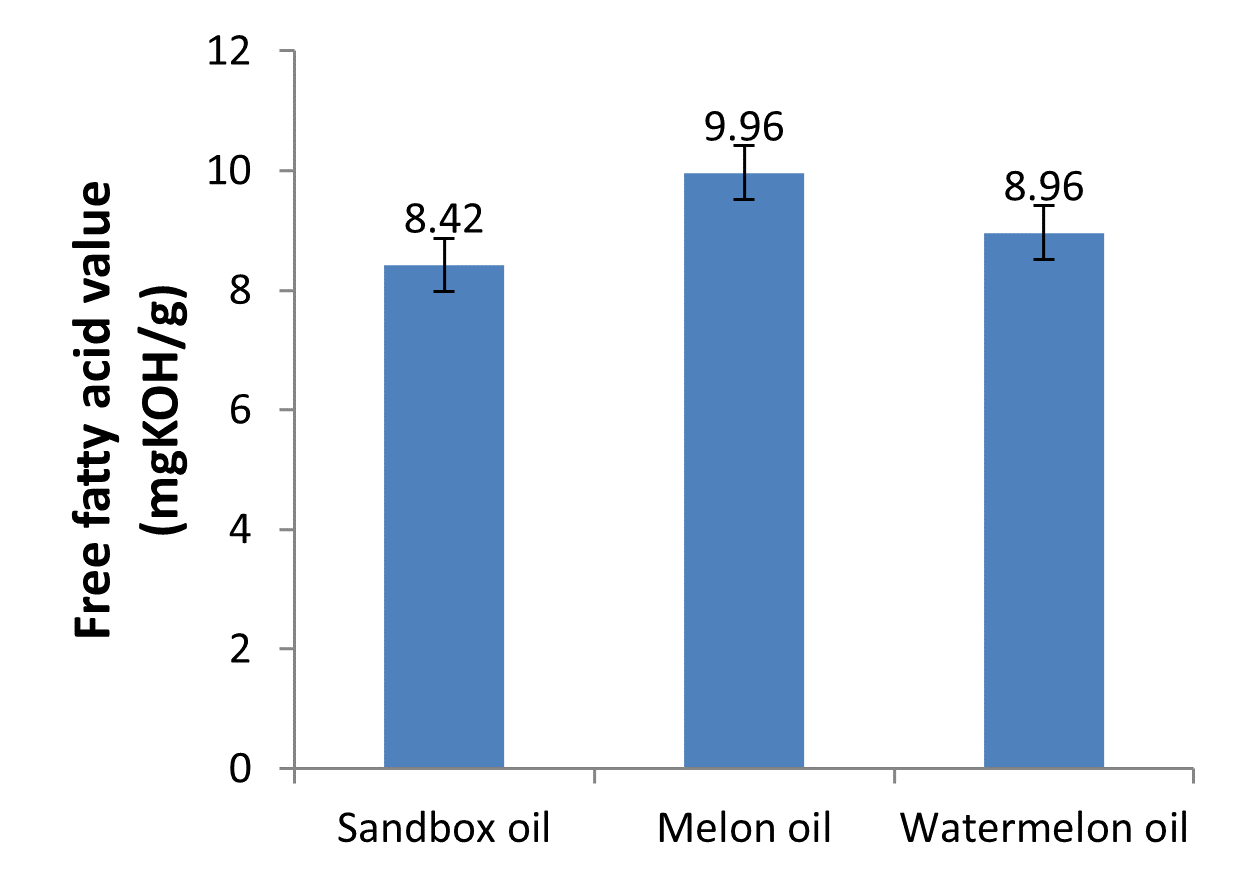
| Free fatty acid (mgKOH/g) | 8.42c | 9.96a | 8.96b |
| Peroxide value (meqKOH/g) | 0.88c | 1.25a | 1.04b |
| Iodine value (gI₂/100g) | 177.66a | 135.24c | 162.10b |
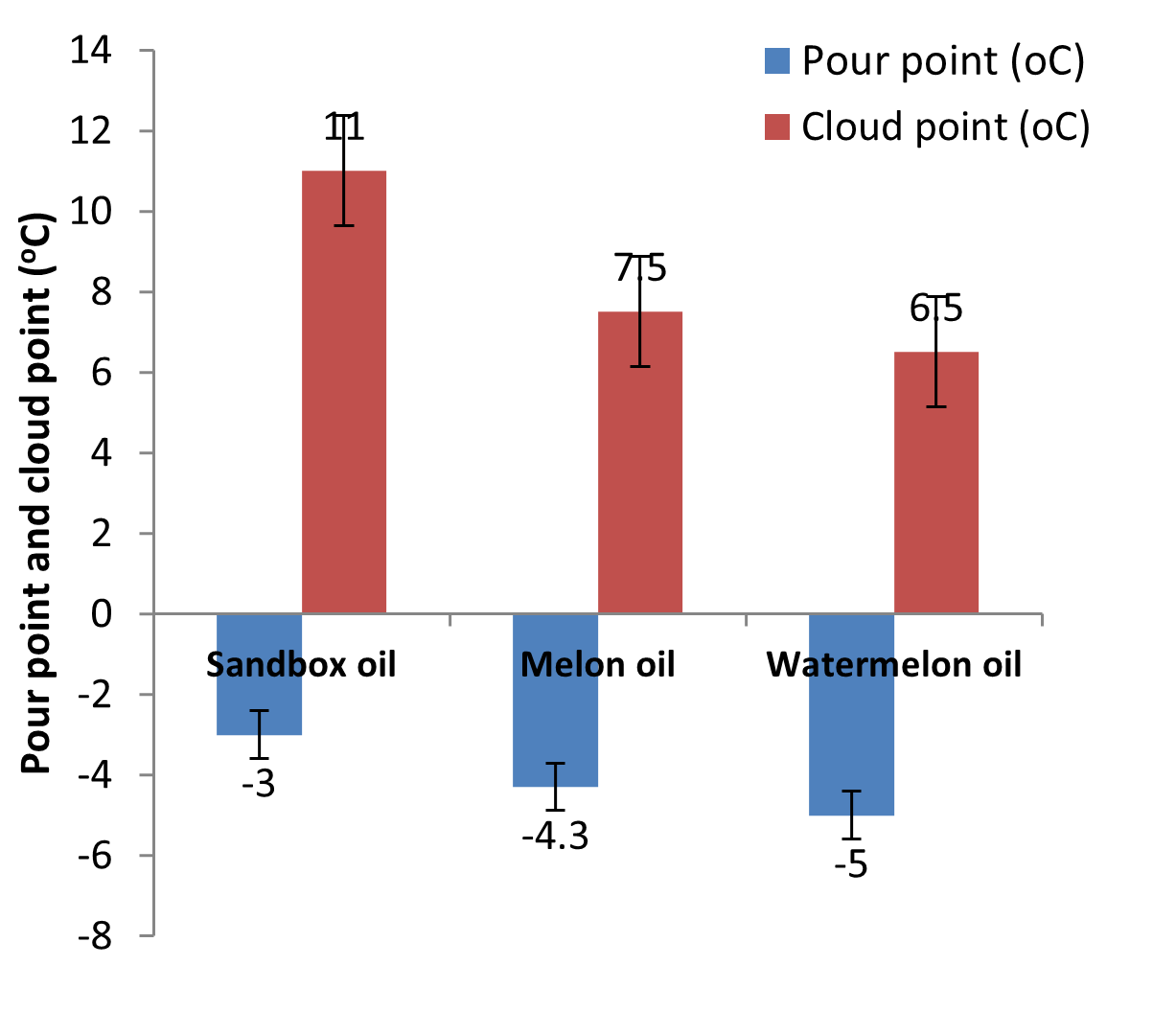
| Pour point (°C) | -3.00a | -4.30a | -5.00b |
| Cloud point (°C) | 11.00a | 7.50b | 6.50b |
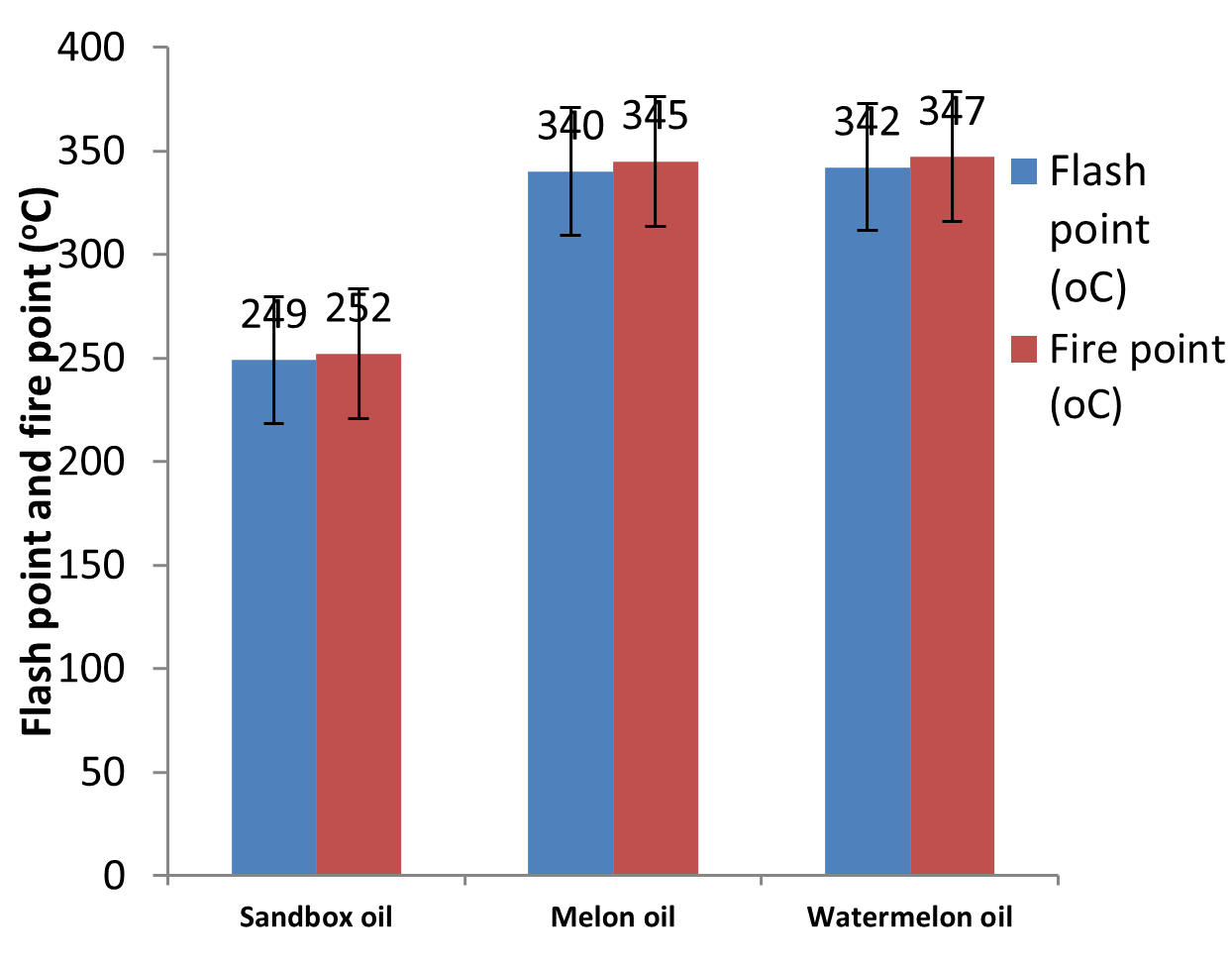
| Flash point (°C) | 249.00b | 340.00a | 342.00a |
| Fire point (°C) | 252.00b | 345.00a | 347.00a |
Values are means of 2 replications. Means with different superscripts within a row are significantly different (p<0.05).
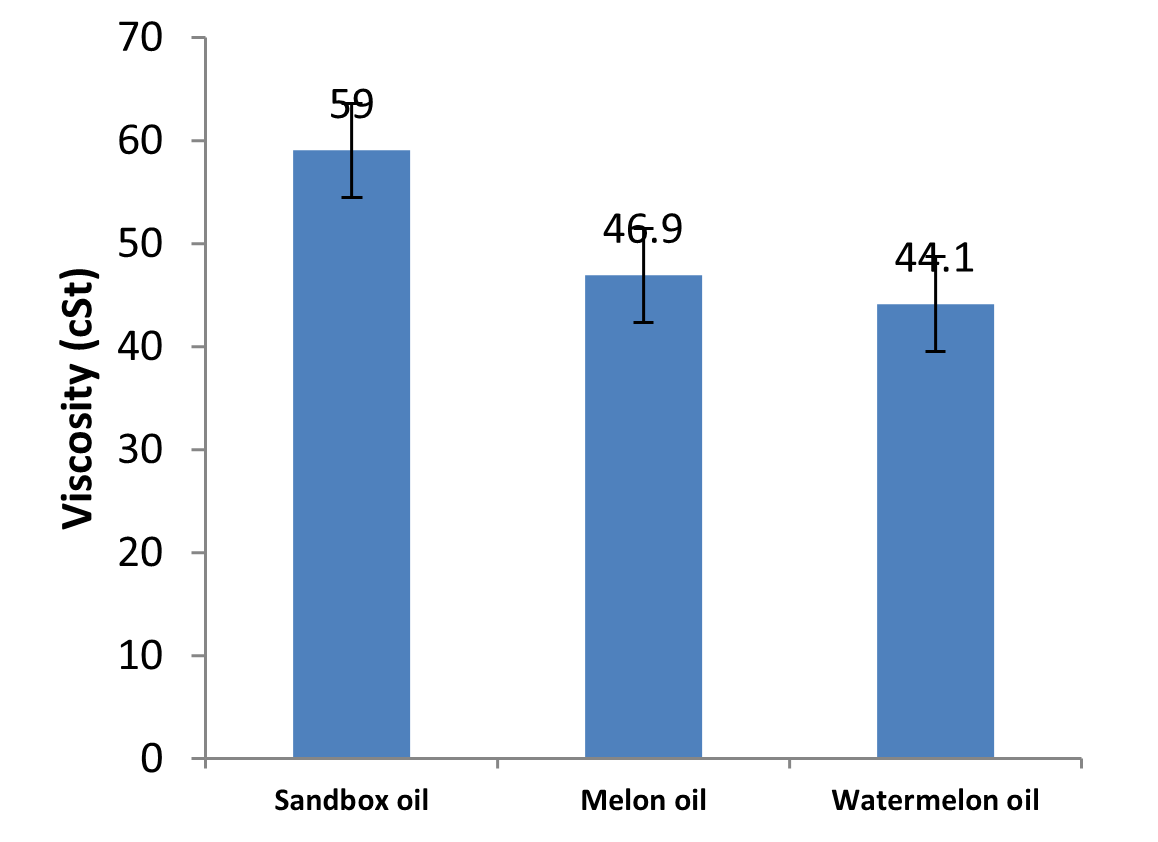
Fig.1: Viscosities of the oils at 28oC
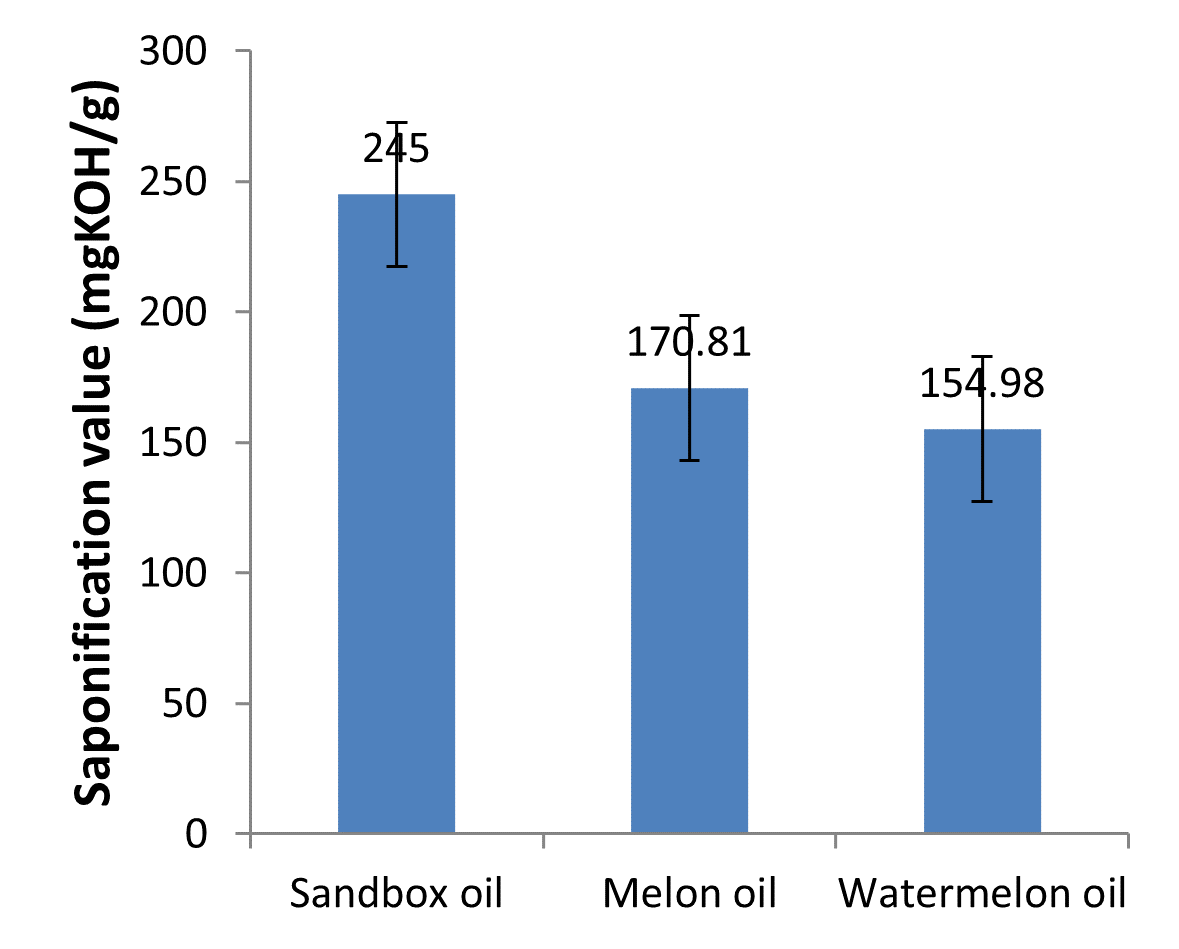
Fig. 2: Saponification values of the oils
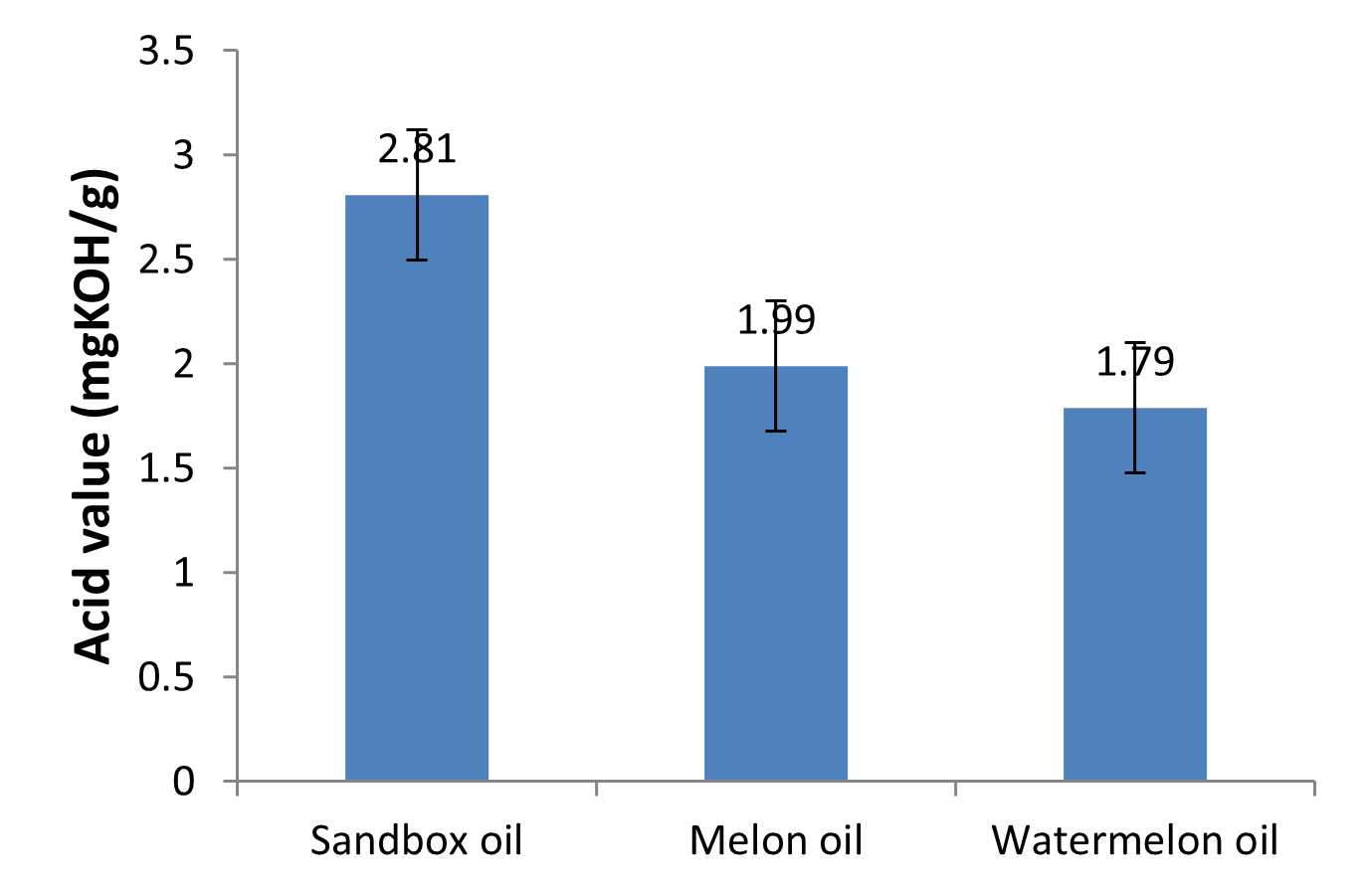
Fig. 3: Acid values of the oils
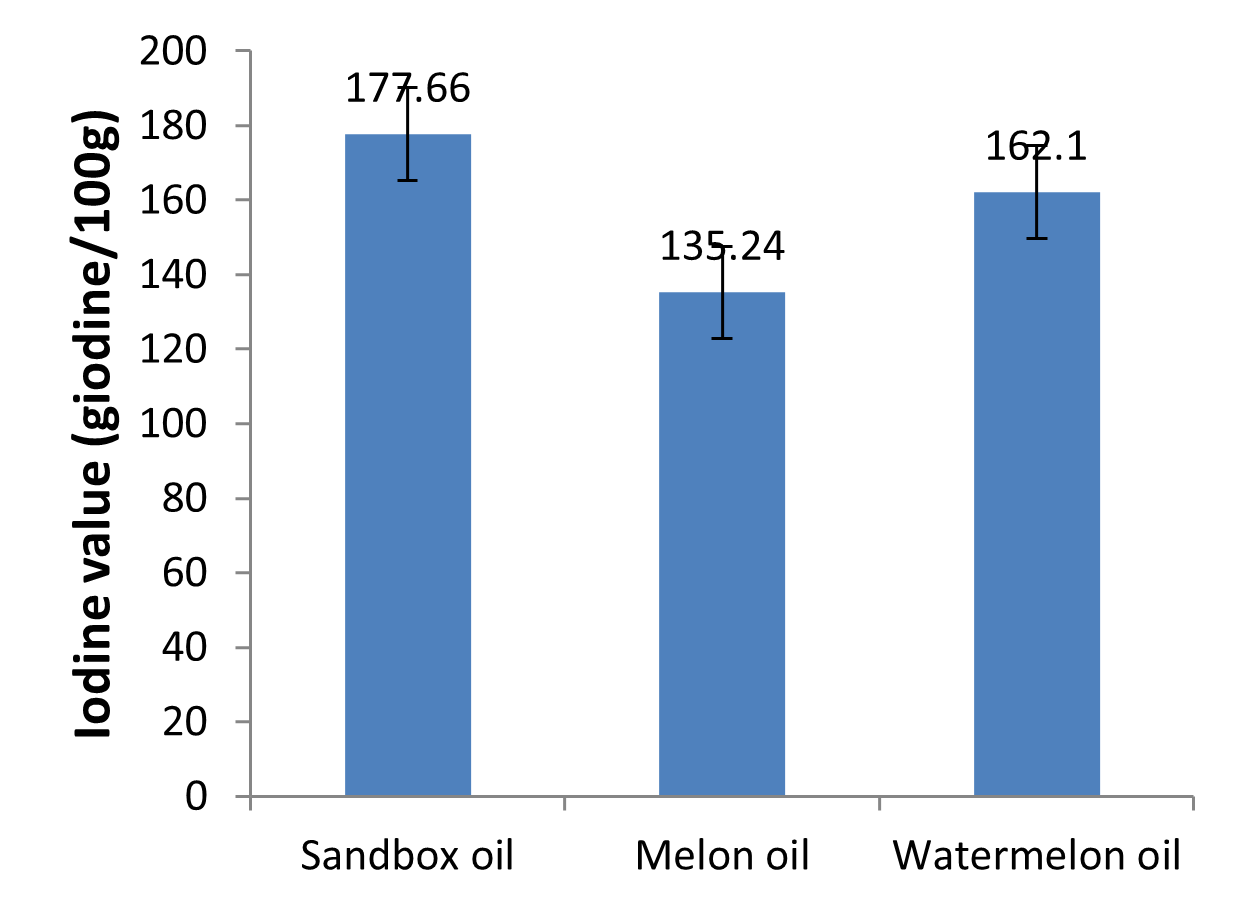
Fig. 4: Iodine values of the oils
Fig. 5: Free fatty acid values of the oils
Fig. 6: Pour and cloud points of the oils
Fig. 7: Flash and fire points of the oils
Moisture content
There was no significant difference (p>0.05) in the moisture contents of the oils as shown in Table 1. The moisture content of the oils was 0.40 % which was within allowable limit of 0.1% – 0.5% for vegetable oils (Okolie et al., 2012). This value shows that the oils will have a longer shelf-stability as it will not easily give in to hydrolysis and ease of attack by oxygen (Nkafamiya, et al., 2010). Sakinah et al. (2016) reported that the presence of water in oil increased the oxidation progress and resulted in premature oil aging. Therefore low moisture content in oil is a requirement for long storage life as oil with low moisture content is expected to be more stable during storage than oils with high values.
Density and Specific gravity
There was significant difference (p<0.05) in densities and specific gravities of the oils as shown in Table 1. According to Fakhri and Qadir (2011), the density of oil is dependent on their fatty acid composition, minor component and temperature. The densities of the oil samples range from 0.82 – 0.90, the densities of the oils falls within the standard range of common oils as approved by the Standard Organization of Nigeria (SON, 2000). The specific gravities of the oil samples range from 0.85 – 0.93, specific gravity is influenced by the chemical composition of the oils. An increase in the amounts of aromatic compound of the oil results in an increase in the specific gravity, while an increase in the saturated compounds results in a decrease in the specific gravity. According to Yahaya et al. (2012), specific gravity is commonly used in conjunction with other figures in assessing the purity of oil. The density and specific gravity values of the oils showed that they are less dense than water. Hence, in case of contamination with water, water will settle below the oil and will be subsequently drained off. Sandbox oil is the densest oil with highest specific gravity when compared with melon and watermelon oils. The specific gravities are in the range found for common oils such as palm oil, olive oil and coconut oil as reported by Aji et al. (2015).
Refractive index
Refractive index of the oil is the degree of refraction of a beam of light that occurs when it passes from one transparent medium to another. It is the ratio of speed of light at a defined wavelength to it speed in the oil. It varies with wavelength and temperature, the degree and type of unsaturation, the type of substitutions of component fatty acids and with accompanying substances. Refractive index indicates the level of saturation of the oil and is widely used in quality control to check for the purity of materials and to follow hydrogenation and isomerisation (Aremu, et al., 2015). There was no significant difference (p>0.05) in the refractive indexes of the oils as shown in Table 1. The refractive indices of the oil samples range from 1.465 – 1.470, which was within the acceptable range of 1.4677 to 1.4707 for virgin, refined and refined-pomace oils as reported by Aremu, et al., (2015). The values showed that the oils are less thick when compared with most oils whose refractive indices were between 1.480 and 1.490 as reported by Olatidoye et al. (2010). These values are satisfactory as they lie within the standard range found in common oils as reported by Aji et al. (2012). The values obtained are also in close agreement with the values obtained by Akintayo et al. (2002) for common oils such as coconut oil and soy bean oil.
Viscosity
Viscosity is one of the critical properties of oil. It is important in the design of process equipment, heat transfer equipment, process piping design and pressure drop determination. There was significant difference (p<0.05) in the viscosities of the oils at room temperature (28oC) as shown in Table 1 and Fig. 1. The viscosities of the oil samples range from 44.10 – 59.00, which are agreement with the viscosity values of some common oils such as soy bean oil, palm oil and coconut oil reported by Olatidoye et al. (2010). The results showed that sandbox oil is more viscous than other oils. The more viscous oil is the better it use for industrial purposes (Aremu et al., 2015). Those oils with low viscosity value indicate that they are light and probably highly unsaturated. Viscosity of oils depends on molecular structure and decreases with the unsaturation of fatty acids (Kim et al., 2010). Oils are mixtures of triglycerides and their viscosity depends on the nature of the triglycerides present in the oil. The viscosity changed due to the different arrangement of the fatty acids on the glycerol backbone of the triglyceride molecule.
Saponification value
Saponification value is a measure of oxidation during storage and also indicates deterioration of the oils. It enhances the quality of the oil because it shows the presence of lower molecular weight components in 1 gram of the oil which will yield more energy on combustion (Aremu et al., 2015). The saponification value of sandbox oil (245.00 mgKOH/g) was significantly high when compared with the values for melon oil (170.81 mgKOH/g) and watermelon oil (154.98 mgKOH/g) as shown in Table 1 and Fig. 2. These values are in agreement with the saponification values of some common oils such as soy bean oil, palm oil and coconut oil reported by Olatidoye et al. (2010). These values indicate that these oils contained high proportion of free fatty acids. The reason being that saponification value is said to have strong positive correlation with free fatty acids content. It has been reported by Olatidoye et al. (2010) that oils with higher saponification values contain high proportion of free fatty acids. According to Yerima et al. (2018), the higher the free fatty acids, the higher the saponification value and vice versa. The presence of high saponification values which indicate the presence of high percentage of free fatty acids might lead to foam formation. Foaming is a desired characteristic of good surfactants with applications in preparation of emulsions, soaps and detergents formulation (Kudirat and Otutu, 2016). This suggests that these oils can be used in production of soaps, shampoos, lather saving creams, paints and bio-lubricants. There could also be an extension to their application into stable foam by incorporation of other ingredients. Therefore, these oils may enjoy usage in many other industrial applications.
Acid value
Acid value is an important indicator of vegetable oil quality. It is defined as the number of milligrams of potassium hydroxide required to neutralize the free fatty acids present in one gram of fat. It is a relative measure of rancidity as free fatty acids are normally formed during decomposition of oil glycerides. The value is a measure of the amount of fatty acids which have been liberated by hydrolysis from the glycerides due to the action of moisture, temperature and/or lypolytic enzyme lipase. Acid value is used as an indicator for edibility of an oil and it suitability for use in industrial applications (Aremu et al., 2015). There was significant difference (p<0.05) in the acid values of the oils as shown in Table 1 and Fig. 3. The acid value of sandbox oil (2.81mgKOH/g) was higher than that of the melon (1.99 mgKOH/g) and watermelon oils (1.79 mgKOH/g). These values falls within the range of 0.34 – 68.88 reported by Aremu et al. (2015) for common oils such as olive oil, soy bean oil, coconut oil and palm oil. Generally, the acid values of the oils were low which indicates that the oils were in good non-degraded state of the lipids and were within limits for industrially useful oils (Oyedeji and Oderinde, 2006). This is very good because the lower the acid value of the oil, the higher the quality. According to Okolie et al. (2012), oils with low acid value are useful industrially in the manufacture of lubricant, paint and varnish. Low acid values in oils indicate that the oils will be stable over a long period of time and protect against rancidity and peroxidation.
Free fatty acid
Free fatty acid is the value of specified fatty acid in oil. The fatty acid profile of oils plays an important role in their stability and nutritional value. Oils with substantial amounts of unsaturated fatty acids are susceptible to oxidation. Aremu et al. (2015) reported that the quantity of free fatty acid in oil is an indicator of its overall quality; high quality oils are low in free fatty acids. Li et al. (2007) reported that oil with low free fatty acid has lesser susceptibility to rancidity. An excessive amount of free fatty acids lowers the flash point of oil and will cause ‘popping’ of the oil during heating. The free fatty acid concentrations of the oils were low which were consistent with the low acid values obtained. The free fatty acid values of the oils range from 8.42 to 9.96, which are around the free fatty acid values of common oils such as groundnut oil, castor oil and coconut oil reported by Aremu et al. (2015). There was significant difference (p<0.05) in the free fatty acid concentration of the oils as shown in Table 1 and Fig. 5. Melon oil had highest free fatty acid concentration (9.96 mgKOH/g), followed by watermelon (8.96 mgKOH/g), while sandbox oil had the lowest concentration (8.42 mgKOH).
Peroxide value
Peroxide value is the most common indicator of lipid oxidation. High values of peroxide value according to Aremu et al. (2015) indicates high levels of oxidative rancidity of the oils and also suggest absence or low levels of antioxidant. The peroxide value of the oils were found to be at the range of 0.88 – 1.25 meqKOH/g, which is low and falls below the standard range of 2 – 10 meqKOH/g reported by Saeed and Shola (2015). Sandbox oil had lowest peroxide value (0.88 meqKOH/g) when compared with melon oil (1.25 meqKOH/g) and watermelon oil (1.04 meqKOH/g). There was significant difference (p<0.05) in the peroxide values of the oils as shown in Table 1. These low peroxide values indicate that the oils are stable to oxidative rancidity and can stand oxidative degradation.
Iodine value
The iodine value could be used to quantify the amount of double bond present in the oil which reflects the susceptibility of the oil to oxidation. It is the measurement of fats and oils unsaturation. High iodine value means high degree of unsaturation fats and oils. The higher the iodine value, the less stable, softer, more reactive and susceptible to oxidation the oil will be. Aremu et al. (2015) reported that the lower the iodine value the lesser the number of unsaturated bonds; thus the lower the susceptibility of such oil to oxidative rancidity. Oils with high iodine value according to Yerima et al. (2018) have lower melting point and performs better in cold weather. There was significant difference (p<0.05) in the iodine values of the oils, with sandbox oil having highest value (177.66 gI2/100g), followed by watermelon (162.10 gI2/100g), and melon (135.24 gI2/100g) as shown in Table 1 and Fig. 4. These values compete favourably with the iodine values of some common oils such as olive oil, soy bean oil, palm oil and coconut oil reported by Olatidoye et al. (2010). These iodine values suggested that the oils are good drying oil; a good drying oil should have iodine value of 130 and above (Aremu et al., 2015). The iodine value of the oils was high, owing to the fact that the oils contain unsaturated glycerides, which have the ability to absorb a definite amount of iodine and can be used for the production of lubricant, paint and alkyd resin. According to Wara et al. (2011), oils whose iodine value is between 100 – 150 possess the property of absorbing oxygen on exposure to the atmosphere, become thicken and remain sticky but do not form a hard dry film. They can be used in the production of margarine and soap.
Pour and cloud points
Pour point is the lowest temperature at which movement was observed while cloud point is the temperature at which a cloud of crystals first appear when the lubricant is cooled. Pour points of the oils range from -3oC to -5.00oC; while cloud points of the oils range from 6.50oC to 11.00oC as shown in Table1 and Fig. 6. These values are agreed with the pour and points values of some common oils such as groundnut oil, soy bean oil, jatropha oil and coconut oil reported by Olatidoye et al. (2010). Low temperature fluidity according to Sripada (2012) is the most essential property for oils to perform in environments that are extremely cold.
Flash and fire points
The flash point is the temperature at which an instantaneous flash occur when flame is brought near the oil while the temperature at which the vapour of the oil burns constantly for 5 seconds is the fire point. Fire point is always flash point plus 5oC up to 400oC. The flash points of the oils range from 249oC to 342oC, while the fire points range from 252oC to 347oC as presented in Table 1 and Fig. 7. It is clear from the results obtained that the oils have very good flash and fire points as they can be used in both humid and temperate regions and transported safely with minimum risks of explosion. These values are similar to the values reported by Ettefaghi et al. (2013) for common oils like jatropha oil, olive oil and coconut oil.
The phytochemical composition of oils from sandbox seed, melon and watermelon seed are shown in Table 2 and Fig. 8.
Table 2: Quantitative composition of phytochemical in the oils from sandbox seed, melon and watermelon seed
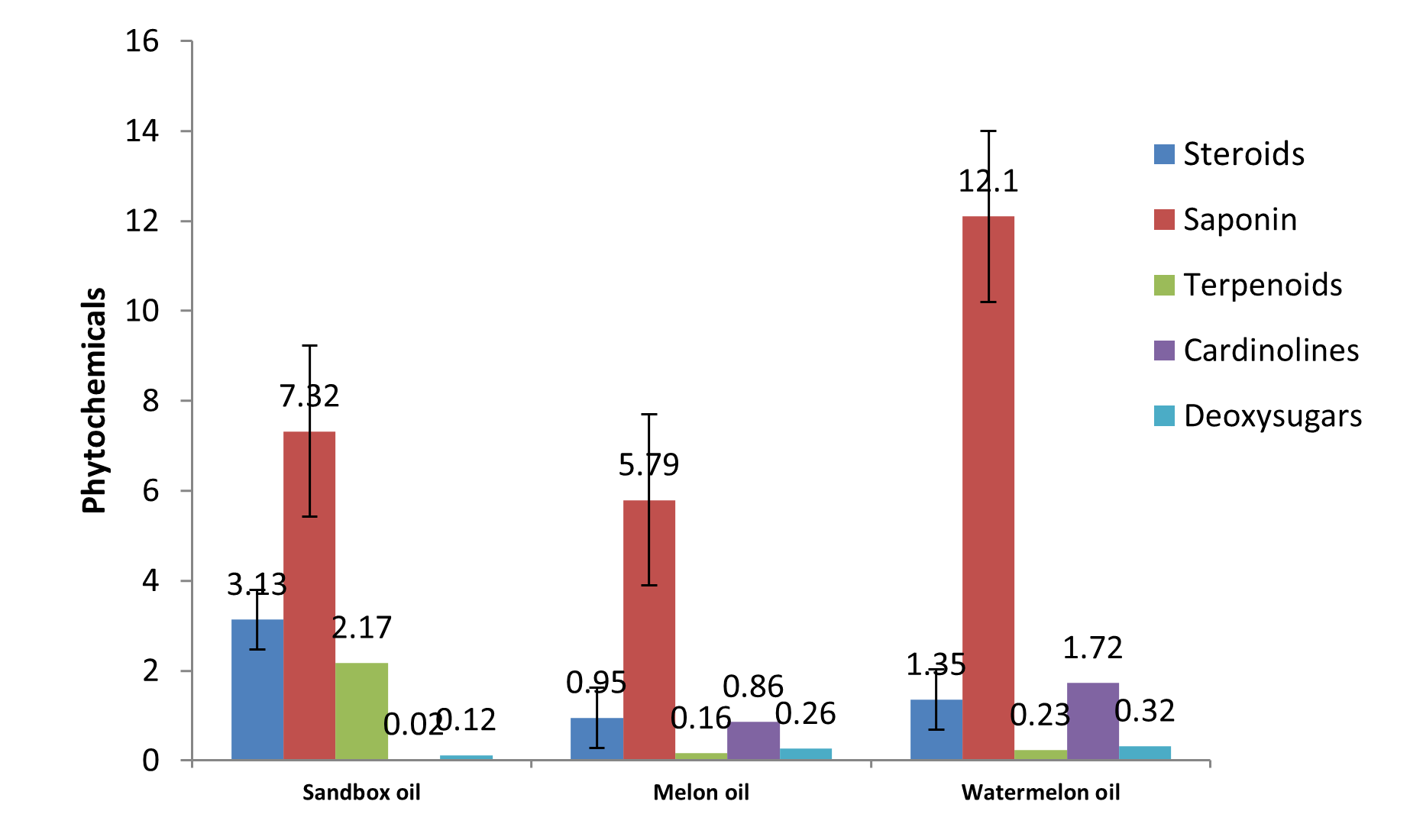
| Phytochemicals | Sandbox oil | Melon oil | Watermelon oil |
| Steroids | 3.13a | 0.95c | 1.35b |
| Saponin | 7.32b | 5.79c | 12.10a |
| Terpenoids | 2.17a | 0.16b | 0.23b |
| Cardinolines | 0.02c | 0.86b | 1.72a |
| Deoxysugars | 0.12c | 0.26b | 0.32a |
Values are means of 2 replications. Means with different superscripts within a row are significantly different (p<0.05).
Fig. 8: Phytochemical composition of the oils
The phytochemical screening of oils revealed the presence of saponins, deoxysugars, terpenoids, and cardenolides and steroids.
Steroids
Plant steroids (or steroid glycosides) also referred to as ‘cardiac glycosides’ are one of the most naturally occurring plant phytoconstituents that have found therapeutic applications as arrow poisons or cardiac drugs (Firn, 2010). The cardiac glycosides are basically steroids with an inherent ability to afford a very specific and powerful action mainly on the cardiac muscle when administered through injection into man or animal. Steroids (anabolic steroids) have been observed to promote nitrogen retention in osteoporosis and in animals with wasting illness (Madziga et al., 2010). Diosgenin and cevadine (from Veratrum veride) are examples of plant steroids. Sandbox oil had highest steroids content of 3.1 g/100g; melon oil had 0.95 g/100g, while watermelon oil had 1.35 g/100g as presented in Table 2 and Fig. 8. These values are in agreement with the steroid values of some common oils reported by Kudirat and Otutu (2016).
Saponins
Saponins are mostly amorphous in nature, they are soluble in water and alcohol but insoluble in non-polar organic solvents such as ether, benzene and n-hexane, and like glycosides on hydrolysis, they give aglycones. They possess ‘soaplike’ behaviour in water, i.e. they produce foam. They possess a bitter and acrid taste, besides causing irritation to mucous membranes. Saponins are also important therapeutically as they are shown to have hypolipidemic and anticancer activity. Saponins are also necessary for activity of cardiac glycosides. Saponins have the property of precipitating and coagulating red blood cells. Some of the characteristics of saponins include formation of foams in aqueous solutions, haemolytic activity, cholesterol binding properties and bitterness (Rita et al., 2015). In humans, saponins might fight cancer and infection (Kudirat and Otutu, 2016). There was significant difference (p<0.05) in the contents of saponins in the oils as presented in Table 2 and Fig. 8. Saponins content of sandbox oil was 7.32 g/100g; melon oil had 5.79 g/100g, while watermelon oil had the highest content of 12.10 g/100g. The values compete favourably with the saponin values of common oils reported by Kudirat and Otutu (2016).
Terpenoids
Terpenoids are among the most widespread and chemically diverse groups of natural products. They are flammable unsaturated hydrocarbons, existing in liquid form commonly found in essential oils, resins or oleoresins (Firn, 2010). Terpenoids includes hydrocarbons of plant origin of general formula (C5H8)n and are classified as mono-, di-, tri- and sesquiterpenoids depending on the number of carbon atoms. Examples of commonly important monoterpenoids include terpinen-4-ol, thujone, camphor, eugenol and menthol. Diterpenoids (C₂0) are classically considered to be resins and taxol, the anticancer agent, is the common example. The common triterpenoids include amyrins, ursolic acid and oleanic acid. Monoterpenoids are major components of many essential oils (Martinez et al., 2008). Terpenoids are antifungal and antibacterial which is attributed to their membrane disruption action and inhibitory action on bacterial cell or fungus (Tawheed and Monika, 2014). Sandbox oil had highest terpenoids content of 2.17 g/100g; melon oil had 0.16 g/100g, while watermelon oil had 0.23 g/100g as presented in Table 2 and Fig. 8. The values are similar to the terpenoids values of some common oils reported by Kudirat and Otutu (2016).
Deoxysugar
Deoxysugar is a form of glycosides having a hydroxyl group replaced with a hydrogen atom. Glycosides are colorless, crystalline carbon, hydrogen and oxygen-containing (some contain nitrogen and sulfur) water-soluble phytoconstituents, found in the cell sap. Chemically, glycosides contain a carbohydrate (glucose) and a non-carbohydrate part (aglycone or genin) (Firn, 2010). Alcohol, glycerol or phenol represents aglycones. Glycosides are neutral in reaction and can be readily hydrolyzed into its components with ferments or mineral acids.
Deoxysugar content of in sandbox seed oil was 0.12 g/100g; melon oil had 0.26 g/100g, while watermelon oil had 0.32 g/100g as presented in Table 2 and Fig. 8. These values are in agreement with the deoxysugar values of some common oils reported by Kudirat and Otutu (2016).
CONCLUSION
This study indicated that sandbox, melon and watermelon oils are rich in important properties. The physicochemical properties of the oils indicated that the oils may be used industrially for the production of bio-lubricants, grease, paints, soaps, shampoos and lather saving creams. The oils could be useful in emulsifier formulation, deep frying, and baking if subjected to further refining.
These oils also contain phytochemical such as saponin, terpenoid, cardinolides, steroids and deoxysugars which are known to have great health significance. The presence of these phytochemicals in the oil has high nutritional importance in diet and to human health, which aid in the prevention and protection against cancer and inflammation. These compounds have proven to have medicinal properties including anticancer, antitumor, anti-malarial, anti-diuretic, antimicrobial, antifungal activities among others. Oils from melon and watermelon seeds have lower acid value, which means it can be used as a good source of edible oil. These oils have great potentials as nutritionally promising oil for a variety of industrial applications in the production of food emulsions. Therefore the utilization of sandbox and watermelon seeds should be encouraged to reduce dumping and environmental hazard.
REFERENCES
- Abere, J.O. (2017). Improved performance of bio-lubricant by nanoparticles additives. Unpublished PhD Thesis, Department of Mechanical Engineering, University of Sheffield.
- Achu, M.B., Fokou, E., Tchiegang, C., Fotso, M and Tchouanguep, F.M. (2005). Nutritive values of some cucurbitaceae oilseeds from different regions in Cameroon. Afr. J. Biotechnol. 11:1329–1334.
- Adewuyi, A., Awolade, P.O. and Oderinde, R.A. (2014). Hura crepitans seed oil: An alternative feedstock for biodiesel production. Journal of Fuels. 2014: 1 – 9.
- Aji, M.M., Kyari, S.A. and Zoaka, G. (2015). Comparative studies between bio-lubricants from jatropha oil, neem oil, and mineral lubricant (Engen Super 20W/50). Applied Research Journal. 1(4): 252 – 257.
- Akintayo, E.T and E. Bayer, (2002). Characterisation and some possible uses of Plukenetia conophora and Adenopus breviflorus seeds and seed oils. Bioresour. Technol, 85:95 – 97.
- Amdebrhan, B.T., Damtew, L., Tesfay, D., Endris, H. and Tekeste, G. (2015). Production of biolubricant from castor (Ricinus) oil. International Journal of Engineering Innovations & Research. 4(5):2277 – 5668.
- Aremu, M.O., Ibrahim, H. and Bamidele, T.O. (2015). Physicochemical characteristics of the oils extracted from some Nigerian plant foods – A Review. Chemical and Process Engineering Research. 32:36 – 52.
- ASTM D1218-01. (2001). Standard test method for determination of the refractive index and refractive dispersion of hydrocarbon liquids. American Society for Testing Materials International, West Conshohocken, USA.
- ASTM D445-15a (2015). Standard test method for kinematic viscosity of transparent and opaque liquids (and the calculation of dynamic viscosity). American Society for Testing Materials International, West Conshohocken, USA.
- ASTM D93-02a. (2003). Standard test methods for flash point by Penky-Martens closed cup tester. American Society for Testing Materials International, West Conshohocken, USA.
- ASTM D97-05. (2005). Standard test method for pour point of petroleum products. American Society for Testing Materials International, West Conshohocken, USA.
- Baboli, Z. M. and Kordi, A. A. S. (2010). Characterisation and consumption of watermelon seed oil and solvent extraction parameters effects. Journal of the American Oil Chemists’ Society, 87:667 – 671.
- Bueno, J.M., Saez-plaza, P., Ramos-escudero, F., Jimenez, A.M., Fett, R. and Asuero, A.G. (2012). Analysis and antioxidant capacity of anthocyanin pigments. Part II: Chemical structure, colour, and intake of anthocyanins. Critical Reviews in Analytical Chemistry, 42: 126 – 151.
- De Conto, L. C., Gragnani, M. A. L., Maus, D., Ambiel, H. C. I., Chiu, M. C., Grimaldi, R. and Goncalves, L. A. G. (2011). Characterisation of crude 62 watermelon seed oil by two different extraction methods. Journal of American Oil Chemists’ Society. 88:1709 – 1714.
- Ettefaghi, E., Ahmadi, H., Rashidi, A., Mohtasebi, S. and Alaei, M. (2013). Experimental evaluation of engine oil properties containing copper oxide nanoparticles as nanoadditive. International Journal of Industrial Chemistry. 4(28):1 – 6.
- Fakhri, N.A and Qadir, H.K. (2011). Amino acid contents of foods and biological data on protein. Journal of environmental science and engineering. 5:844 – 849.
- Firn, R. (2010). Phytochemical properties of the seed of watermelon (Citrulluslanatus).Nature’s Chemicals. Oxford University Press. Oxford. 74 – 75.
- Giwa, S.O., Adama, K.O. Nwaokocha, C.N. and Solana, O.I. (2016). Characterization and flow behaviour of Sandbox (Hura crepitans Linn) seed oil and its methyl esters. International Enegry Journal. 16:65 – 72.
- Idowu, D.O., Abegunrin, T.P, Ola, F.A., Adediran, A.A. and Olaniran, J.A. (2012). Measurement of some engineering properties of sandbox seeds (Hura crepitans). Agric. and Biology Journal of North America. 3(8):318 – 325.
- Jarret, R.L. and Levy, I.J. (2012). Oil and fatty acid content in seed of Citrullus lanatus Schard. Journal of Agricultural and Food Chemistry. 60:5199 – 5204.
- Kim, J., Kim, D.N., Lee, S.H., Yoo, S.H. and Lee, S. (2010). Correlation of fatty acid composition of vegetable oils with rheological behaviour and oil uptake. Food Chem., 118: 398 – 402.
- Kudirat, T.S. and Otutu, O.L. (2016). Phytochemical composition and radical scavenging activities of watermelon (Citrullus lanatus) seed constituents, Croat. J. Food Sci. Technol. 8 (2): 8 – 62.
- Latha, S. (2012). Fatty Acid Profile of Unconventional Oilseeds. Advances in Food and Nutrition Research, 67:141 – 184.
- Li, S.G., Zhang, H. and Xue, W.T. (2007). A novel method for the determination of acid value of vegetable oils. Eur. J. Lipid Sci. Tech., 109:1088 – 1094.
- Mabaleha, M.B., Mitei, Y.C. and Yeboah, S.O. (2007). A comparative study of the properties of selected melon seed oils as potential candidates for development into commercial edible vegetable oil. J. Am. Oil Chem. Soc. 84, 31–36.
- Madziga, H.A., Sanni, S. and Sandabe, U.K. (2010). Phytochemical and Elemental Analysis of Acalypha wilkesiana Leaf. Journal of American Science. 6 (11):510 – 514.
- Martinez, M.J.A., Lazaro, R.M., del Olmo, L.M.B and Benito, P.B. (2008) Anti-infectious activity in the anthemideae tribe. In: Atta-ur- (Ed.) Studies in Natural Products Chemistry, 35: 445 – 516.
- Mohammed, R.F. and Jorf-Thomas, M. (2003). Determination of the lipid classes and fatty profile of Niger seed (Guizotia abyssinica Cass). Phytochem. Anal. 14:366 – 370.
- Naik, A.U. and Galhe, D.S. (2016). Review on vegetable oil as bio-lubricant. Int. Journal for Scientific Research & Development. 5(10):587 – 589.
- Nkafamiya, I.I., Maina, H.M., Osemeahon, S.A and Modibbo, U.U. (2010). Percentage yield and Physicochemical properties of different groundnut species (Arachis hypogeal). African Journal of Food Science. 4 (7): 418 – 421.
- Ntui, V.O., Thirukkumaran, G., Iioka, S. and Mii, M. (2009). Efficient plant regeneration via organogenesis in ‘‘egusi’’ melon (Colocynthis citrullus L.). Scientia Hort, 119: 397–402.
- Nweze, E.L., Okafor, J.L and Njoku, O. (2004). Antimicrobial Activityies of Methanolic extracts of Trume guineesis (Scchumn and Thorn) and Morinda lucinda used in Nigerian Herbal Medicinal practice. Journal of Biological Research and Biotechnology. 2 (1):34 – 46.
- Oluba, M.O., Ogunlowo, Y.R., Ojieh, G.C., Adebisi, K.E., Eidangbe, G.O and Isiosio, I.O. (2008). Physicochemical properties and fatty acid composition of citrullus lanatus (Egusi melon) seed oil. J. Biol. Sci. 2008 (8): 814–817.
- Okolie, P.N., Uaboi-Egbemi, P.O. and Ajekwene, A.E. (2012). Extraction and quality evaluation of sandbox tree seed (Hura crepitan) oil. World Journal of Agricultural Sciences. 8(4):359 – 365.
- Olatidoye, O.P., Adeleke, A.E., Adegbite, S.A. and Sobowale, S.S. (2010). Chemical composition and nutritional evaluation of sandbox (Hura crepitans) seed flour for domestic consumption and Industrial utilization in Nigeria. Journal of Medical and Applied Bioscience. 2:72 – 83.
- Oniya, O.O., Akande, A.A., Adedeji, A.A. and Olukayode, O.L. (2014). Transesterification of Hura crepitans oil for biodiesel production. Journal of Engineering and Applied Scientific Research. 6(1):91 – 97.
- Oniya, O.O., Oyelade, J.O., Ogunkunle, O. and Idowu, D.O. (2017). Optimization of solvent extraction of oil from sandbox kernels (Hura crepitans L.) by a response surface method. Energy and Policy. 4(1):36 – 43.
- Oyedeji, F.O. and R.A. Oderinde. (2006). Characterization of isopropanol extracted vegetable oils. Journal of Applied Science. 6(11):2510 – 2513.
- Rita N., Baruah, K.K., Sarma, S., Bhuyan, R., Roy, D.C and Mithu, D. (2015). Phytochemical screening of different plants of North-Eastern region of India. J Biosci Bioeng, 2:9 – 11.
- Saeed, M.D. and Shola, E.A. (2015). Extraction and physicochemical properties of some edible seed oil sample in Kano metropolis, Kano state. Bayero Journal of Pure and Applied Science, 8 (2):239 – 244.
- Sakinah, M.H., Amirruddin, A.K., Kadirgama, K., Ramasamy, D., Rahman, M.M. and Noor, M.M. (2016). The application of response surface methodology in the investigation of the tribological behaviour of palm cooking oil blended in engine oil. Advances in Tribology. 2016: 1 – 11.
- Sergent, T., Piront, N., Meurice, J., Toussaint, O. and Scheinder, Y.J. (2010). Anti-inflammatory effects of dietary phenolic compounds in an in vitro model of inflamed human intestinal epithelium. Chemico-Biological Interactions. 188:659 – 667.
- Snyder, S.M., Reber, J.D., Freeman, B.L., Orgad, K., Eggett, D.L. and Parker, T.L. (2011). Controlling for sugar and ascorbic acid, a mixture of flavonoids matching navel oranges significantly increases human postprandial serum antioxidant capacity. Nutrition Research, 31: 519 – 526.
- SON (2000). Standards for edible refined palm oil and its processed form. Standard Organization of Nigeria, Nigeria.
- Sripada, P.K. (2012). Comparative tribological property evaluation of trimethylolpropane based bio-lubricants derived from methyl oleate and canola biodiesel. Unpublished M.Sc. Thesis, Department of Chemical and Biological Engineering, University of Saskatchewan, Canada.
- Tanaka, T., Shnimizu, M and Moriwaki, H. (2012). Cancer chemoprevention by carotenoids.Molecules, 17: 3202 – 3242
- Tawheed, A. and Monika, T. (2014). A comparative study on proximate composition, phytochemical screening, antioxidant and antimicrobial activities of Linum usitatisimum L. (flaxseeds). Int. J. Curr. Microbiol. App. Sci., 3(4):465 – 481.
- Wara, A.A., Wawata, I.G., Gunu, S.Y. and Aujara, K.M. (2011). Extraction and physicochemical analysis of some selected Northern Nigerian industrial oils. Archive of Applied Research, 3 (4):536 – 541.
- Yadav, S., Tomar, A.K., Jithesh, O., Khan, M.A., Yadav, R.N., Srinivasan, A and Yadav, S. (2011). Purification and partial characterization of low molecular weight vicilin-like glycoprotein from the seeds of Citrullus lanatus. The Protein Journal. 30 (8):575 – 580.
- Yahaya, A.T., Taiwo, O., Shittu, T.R., Yahaya, L.E and Jayeola, C.O. (2012). Investment in cashew kernel oil production; cost and return analysis of three processing methods. American Journal of economics, 2 (3):45 – 49.
- Yerima, E.A., Yebpella, G.G., Ogah, E., Longbap, B.D. and Johnson, S.N. (2018). Effect of extraction methods on the physicochemical properties of palm kernel oil. International Journal of Modern Chemistry. 10(1): 104 – 116.