Chitosan Based Copper (II) and Cadmium Nanoparticles: Synthesis, Characterization, and Investigation of Antibacterial Activity
- Gazi Jahirul Islam
- Shaheda Zannah
- Md. Mohinuddin
- 543-561
- Jul 21, 2024
- Materials Science
Chitosan Based Copper (II) and Cadmium Nanoparticles: Synthesis, Characterization, and Investigation of Antibacterial Activity
Gazi Jahirul Islam1*, Shaheda Zannah2 and Md. Mohinuddin1
1Department of Chemistry, University of Barishal,
Barishal-8254, Bangladesh
2Department of Pharmacy, Southeast University,
Dhaka-1208, Bangladesh
*Corresponding Author
DOI: https://doi.org/10.51244/IJRSI.2024.1106042
Received: 24 May 2024; Accepted: 20 June 2024; Published: 21 July 2024
ABSTRACT
Development of novel antibacterial material has become one of the prominent research topics as because bacteria becoming more and more resistant with time and it becomes difficult to combat against bacteria with existing drug materials. Chitosan, a naturally occurring biopolymer offers a feasible framework for the manufacture of metal nanoparticles, which has naturally enhanced antibacterial properties. In this research, copper (Cu2⁺) and cadmium (Cd) nanoparticles (NPs) were synthesized utilizing chitosan as a reducing and stabilizing agent. Chitosan based metal nanoparticles were produced via the solution casting process. The parameters and procedures are different from those used in previous studies. The synthesized nanoparticles were characterized using FTIR, TGA, and SEM analysis. The antibacterial capabilities of these nanoparticles were investigated against different strains of bacteria, which showed, enhanced antibacterial activity. Chi-Cu(II) and Chi-Cd NPs have zones of inhibition 30, 35, 36, 33 mm and 35, 31, 31, 38 mm against Pseudomonas aeruginosa, Salmonella bovismorbificans, Salmonella typhi, and Escherichia coli bacteria respectively. Whereas the standard antibiotic kanamycin has zones of inhibition 22, 22, 20, and 20 mm against those bacteria respectively.
Keywords: Chitosan, nanoparticles, FTIR, TGA, SEM, antibacterial activities
INTRODUCTION
Chitosan is a biomaterial that is renewable and is derived from chitin, the second most prevalent biopolymer and semi-synthetic biopolymer [1, 2]. It is also known as β-[1→4]-2-amino2-deoxy-D-glucopyranose, a basic and functional linear polysaccharide [3]. N-deacetylation of chitin yields a copolymer of 2-amino-2-deoxy-D-glucopyranose and β-[1→4]-linked 2-acetamido-2-deoxy-D-glucopyranose, which is then used to make chitosan [4-6]. Chitosan’s aggregation activity [7], emulsification capacity [8], physicochemical [9, 10] and rheological [11, 12] properties are all primarily influenced by its degree of deacetylation (DD) and molecular weight. Chitosan is widely used in different fields, including biomedicine [13-16], pharmaceuticals [17-19], metal chelation [3, 20], food additives [21, 22] and other industrial applications [23-25]. This is due to its unique set of properties including biocompatibility [26], low or no toxicity [27], biodegradability [28], low immunogenicity [29], and antibacterial properties [30]. Present industrial revolution in nanotechnology have a profound impact on the society, the economy, and the wider globe [31, 32]. So, there is a chance for a safe application of an ecologically friendly method of producing nanoparticles in the medical domain [33, 34]. Numerous biological applications have drawn a lot of attention to nanotechnology [35, 36]. In the domains of medicine, pharmacology and agriculture, nanoparticles have proven efficiency in lowering bacterial and fungal infections [37, 38]. Chitosan based nanomaterials have shown excellent potential in the field of biomedical applications as antimicrobial agents, membrane separators, drug delivery, biomolecule sensing materials, and tissue engineering [39-41] etc. Furthermore, chitosan derivatives and Chi-NPs demonstrated outstanding efficacy in the fields of dentistry, ophthalmology, bioimaging, biosensing, and diagnostics [42, 43]. Nanomaterials such as gold, silver, copper, selenium, titanium, zinc oxide, and magnesium oxide have antibacterial activities against human pathogenic bacteria and fungus [44-46]. Multidrug-resistant (MDR) bacteria are becoming more widespread, and this poses an alarming danger to public health [47, 48]. Among those bacteria Klebsiella pneumoni, Escherichia coli, Streptococcus pneumonia, Enterococcus faecalis, and Staphylococcus aureus are widely known. Gram-negative bacteria deploy a variety of defense strategies against the harmful effects of antibiotics, including the generation of degrading enzymes, efflux pumps, and a low permeability outer membrane [49]. By 2050, 300 million premature deaths and up to USD 100 trillion (GBP 64 trillion) in economic losses will be the true cost of antibiotic resistance [50]. A deadly fungus that affects over 1.2 billion people globally causes at least 1.7 million deaths a year [51]. Fungal strains that are resistant to the majority of commercial antifungal medications are a result of the extensive usage of antifungal medicines [52]. Consequently, it is necessary to find and formulate novel antimicrobial agents.
The earliest description of chitosan nanoparticles dates back to 1994, when Ohya and associates [53] suggested the use of emulsified and cross-linked chitosan nanoparticles to deliver the anticancer medication, 5-fluorouracil intravenously. Since then, lots of research have been conducted on these drug-transporting systems, and the original formulation has been used for various purposes, such as, adding active chemicals to toothpastes [54], or modified afterwards, by applying different manufacturing procedures. Furthermore, many researchers have synthesized novel chitosan nanoparticle formulations with secondary matrix-forming components [55, 56]. Several techniques have been developed for synthesis of chitosan nanoparticles including ionic gelation, polyelectrolyte complexation [55, 56], emulsion-droplet coalescence [57], emulsion solvent diffusion [58], reverse micellar approach [59] etc.
Apart from their catalytic, optical, and electrical conducting characteristics, Cu (II) NPs are appealing due to having noteworthy antibacterial and antifungal capabilities [60-62]. Cioffi et al. reported in 2005 [63] the antifungal and antifouling properties of a Cu NP-polymer composite. M. Taner et al. [64] reported that, the synthesis of Ag-Cu nanoalloy and its bactericidal action against Escherichia coli (E. coli). Ag, however, was confirmed to accumulate in the human body over time and found to be poisonous, resulting in a disorder, called, argyria [65]. A number of techniques, including thermal reduction [66], metal vapor synthesis [67], chemical reduction [68], vacuum vapor deposition [69], micro-emulsion technique [70], and laser ablation [71] have been developed recently for the synthesis of Cu NPs. The synthesis of Cd NPs is of tremendous interest because of its significant applications in the fields of photocatalysis, microelectronics, and catalysis etc. [72, 73].
In this research, chitosan based copper (Cu2⁺) and cadmium (Cd) nanoparticles (NPs) were synthesized. Chitosan based metal nanoparticles were produced via the solution casting process. The generated nanoparticles were characterized using FTIR, TGA, and SEM analysis. The antibacterial capabilities of synthesized nanoparticles were investigated against five different strains of gram-negative bacteria for instance, Pseudomonas aeruginosa, Salmonella bovismorbificans, Salmonella typhi, and Escherichia coli.
EXPERIMENTAL
Materials and Methods
Instruments:
Thermal gravimetric analysis (TGA-50, Shimadzu), Scanning electron microscopy (SEM) (JEOL JSM-6490LA), Electric oven (YCO-010 Series), IR (IR Prestige-21, Shimadzu), Electric balance (AND-HR-200, Sonicator (POWERSONIC 603), Furnace (Thermo Scientific Thermolyne, FB1415M), and Electric magnetic stirrer.
Raw Materials, Chemicals and Reagents:
All the reagents were purchased from Sigma-Aldrich Australia Ltd. and used as received unless otherwise indicated. Acetic acid, Copper (ll) sulphate pentahydrate, Cadmium sulphate, Hydrazine hydrate solution, Sodium hydroxide, (E-Merck, Germany). Shrimp shell was collected from the local market.
Preparation of Chitosan:
The waste from dehydrated shrimp shell was obtained from a local commercial shrimp shell processor. Shells were initially washed with water for several times and then sun-dried until they became totally dried. The dry shell was crushed into coarse particles using a centrifugal crushing mill to produce a product with consistent size. Prior to use, dried ground shell were kept at room temperature in opaque plastic bottles. The modified method was utilized to produce chitosan from shrimp shell waste.
Steps of methods:
The following steps were mostly engaged in this process.
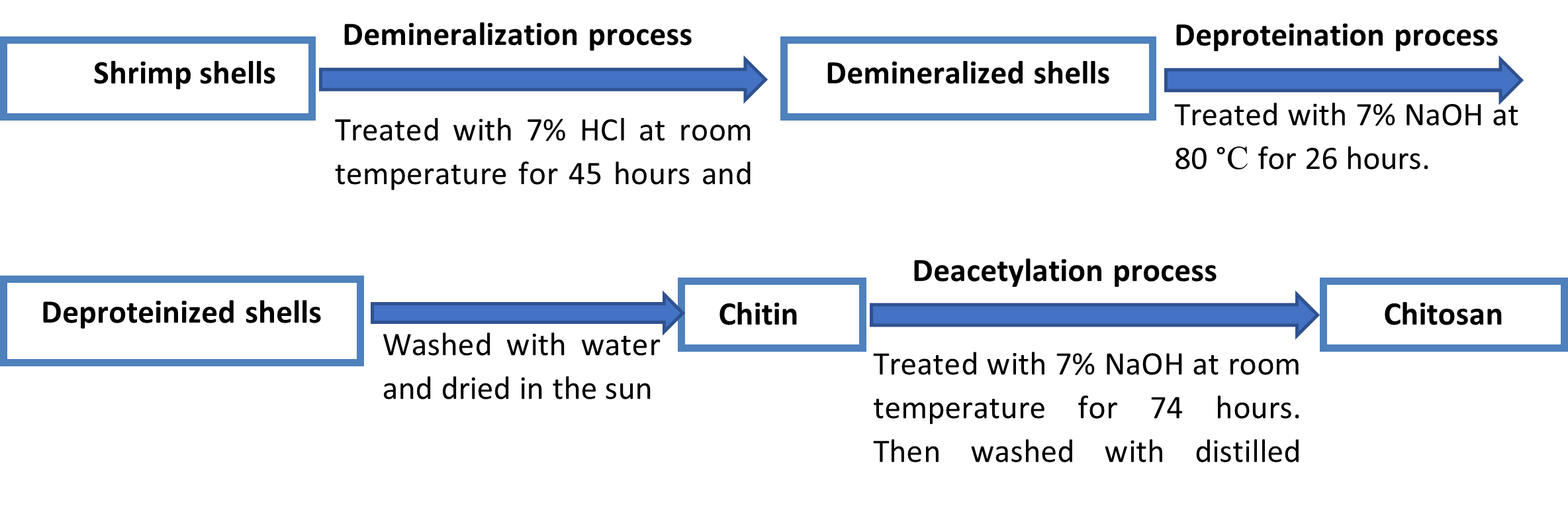
Demineralization of shells: Demineralization is the process of removing minerals, especially calcium carbonate. It could be conducted under the effect of inorganic or organic acids [74, 75]. Demineralization is one of the most important steps in producing chitin and chitosan. It is usually obtained by the inorganic acidic treatment. Shrimp shells have large amounts of minerals which is combined with proteins, and chitin, and the rest of the shell is exoskeleton. Calcium carbonate and calcium phosphate are the main minerals in the shells, which must be discarded to demineralize the shrimp shells [76]. Demineralization is the method of replacing hydroxide (OH–) ions for all anions and hydrogen ions (H+) for all cations. In this phase, the shells were kept in a 1:12 (V/W) ratio of 7% HCl at ambient temperature. After 45 hours the shells were gently squashed and were washed with water to get rid of the calcium chloride and acid.
Deproteinization of shells: The process of deproteinization involves either hydrolyzing or precipitating proteins out of a solution in order to remove them from a biological sample. In order to extract protein, the demineralized shells were treated with 7% NaOH at 80 °C for 26 hours, with solid to solvent ratio of 1:12 (V/W). The residue was then collected and washed under the running tap water until it turned neutral. After washed thoroughly, the product was sun-dried and the final product was chitin.
Production of Chitosan from chitin by deacetylation method: The acetyl groups in the chitin were removed by utilizing a 70% NaOH solution with a solid to solvent ratio of 1:12 (V/W) at room temperature for 74 hours. To ensure a consistent reaction, the mixture was continuously agitated by electrical magnetic stirrer. After that, it was rinsed with distilled water and was cleaned under running tap water. Then it was filtered and oven-dried. Finally, white to off-white flakes of chitosan were generated from shrimp shells.
Flow diagram of the preparation of Chitosan:
Preparation of Chitosan-Cu (II) NPs:
1 g of chitosan was dissolved in 100 mL of 1% acetic acid solution, and 0.8 g of copper(ll) sulfate (CuSO4.5H2O) was added to 100 mL of 1% acetic acid. 20 mL of each solution were mixed and underwent five minutes of sonication. After that, the mixture was magnetically agitated while being heated for roughly 20 minutes to a temperature of between 50 and 80 °C. In this situation, 1.0 M NaOH was gradually and carefully added and mixed into the solution until the blue NPs formation was fully accomplished. It was then filtered, washed many times with distilled water, and dried while being agitated continuously.
Preparation of Chitosan-Cd NPs:
3.8 g of cadmium sulfate was added to 100 mL of 1% acetic acid solution and was sonicated until dissolved. Separately 1 g of chitosan was dissolved in 100 mL of 1% acetic acid solution. 20 mL of each solution were mixed and sonicated for a few minutes. Next, the solution was heated for about 20 minutes at a temperature between 50 and 80 °C while being stirred magnetically. After that, drop by drop of 1 M NaOH solution was added to the mixture until the off-white NPs were formed. Subsequently, it was filtered, and then repeatedly cleaned with distilled water, and then allowed to dry. This is an entirely novel technique for making Cd-NPs based on chitosan.
Moisture Content:
Employing the gravimetric approach, the produced chitosan’s moisture content was ascertained [77]. 2.0 g of the chitosan was weighed out carefully and then heated for 40 minutes at 102 °C. Then it was allowed to cool to room temperature for 10 minutes in a desiccator. It was immediately weighted, and the following formula was used to determine the percentage of the moisture content.
Ash Content:
2.0 g of chitosan sample was added to a crucible that had previously been ignited, cooled, and tared in order to calculate the ash value of the material [78]. The sample was heated for four hours at 650 °C in a muffle furnace. After being allowed to cool to a temperature of less than 200 °C in the furnace, the crucibles were put into desiccators that had a vented top.
Solubility Test:
The solubility of chitosan was checked in a variety of mineral acids, organic acids, and other chemical solvents. It has been found that chitosan was soluble in 1% CH3COOH solution.
FTIR Test:
Infrared spectra were obtained in range of the 4500 – 400 cm-1 by using an FTIR spectrophotometer (IR Prestige – 21, Shimadzu). The samples were placed in an agate mortar, ground to a fine powder using potassium bromide, and then placed in a mini-disc holder, where an automatic hydraulic press created a disc. The machine’s sample cavity held the KBr disk in place. The polystyrene film’s 1601.8 cm-1 peak served as the calibration point for the spectra. Based on a few standard books, provisional band assignments for the significant infrared bands of the different complexes and the relevant ligands have been determined empirically [79, 80].
Scanning electron microscope (SEM) of the Nanoparticles:
The synthesized Chi-NPs were subjected to surface analyses with a Carl Zeiss Scanning Electron Microscope (Germany). After applying 10 µL of the diluted suspension to an aluminum stub, the NPs suspension was dried in a desiccator for a minimum of 24 hours to obtain a fine film. The created NP film was sputter-coated with a gold layer under ultra-high vacuum. SEM photos were obtained with EVO 18 microscopes using smart SEM software.
Thermal gravimetric analysis (TGA):
The thermal analyses were obtained by TA Instruments – 2100 thermal analysis system [79]. Each sample was precisely weighed between 16 and 30 mg, and then placed into a 100 µL metal crucible. It was then placed inside the furnace. A pinhole was pierced into the crucible cover, and the crucible was hermetically sealed.
Antibacterial Activity:
Gram-negative Pseudomonas aeruginosa, gram-negative Salmonella bovismorbificans, gram-negative Salmonella typhi, and gram-negative Escherichia coli were the pathogens against which the complexes’ antibacterial activity were evaluated. The activities were conducted using the disc diffusion technique [81, 82]. Every disk was placed on bacteria-infected plates containing 30 mg of tested substance. The growth of inhibition results were compared with the standard antibiotic kanamycin.
RESULTS AND DISCUSSION
Characterization of Chitosan:
Moisture content:
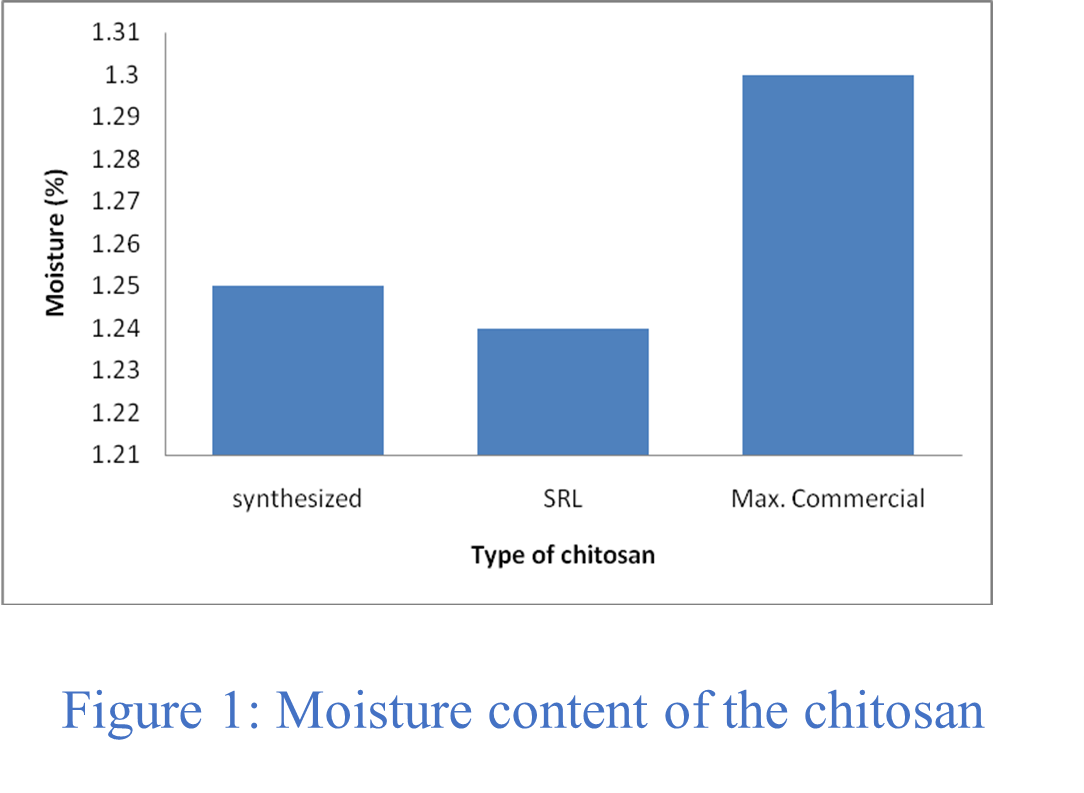
Table 1 displays the physio-chemical and functional properties of the produced chitosan. The exceptionally pure chitosan was contrasted with the commercially manufactured chitosan made from chitin and shrimp shell. Typically, the moisture content of commercial chitosan ranges from 1.00 to 1.30%, depending upon the time of the year, relative humidity, and solar radiation intensity, etc. The moisture content of prepared chitosan was found to be 1.25%, with a good agreement with the commercial extra pure chitosan, which has moisture content apprximately 1.24% (Figure 1).
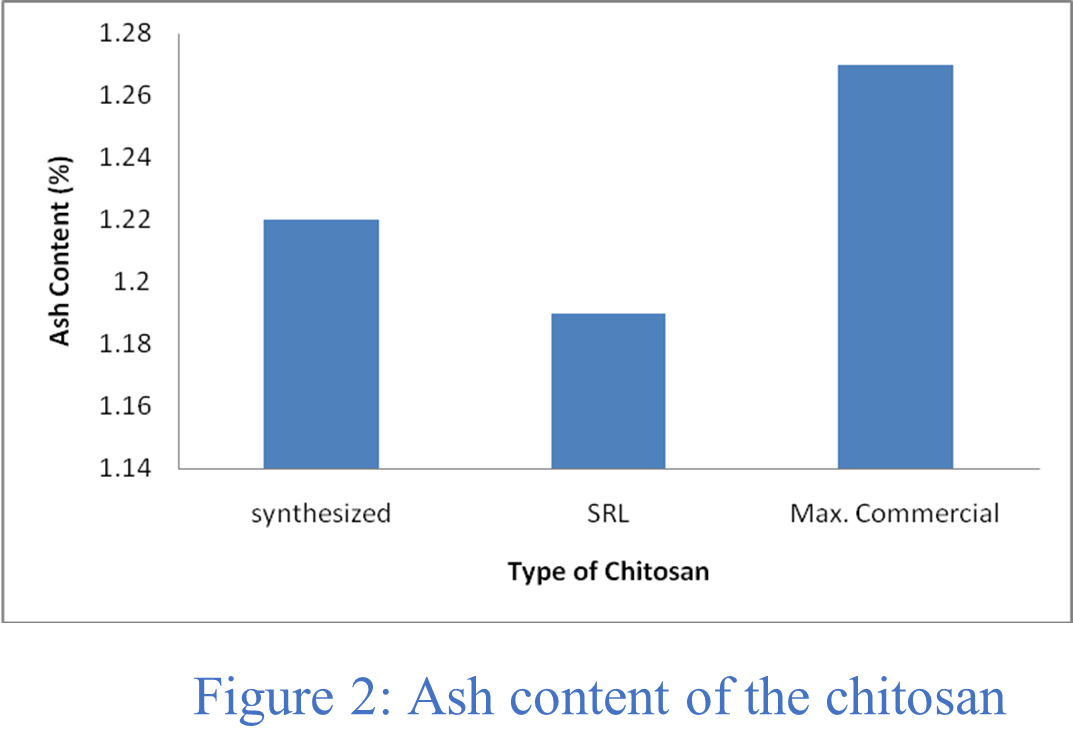
Table 1: Solubility, Moisture and Ash content of chitosan.
| Obs. No. | Test Parameters | Synthesized % | Standard (SRL) % | Commercial % |
| 1 | Moisture | 1.25 | 1.24 | 1.00 – 1.30 |
| 2 | Ash | 1.22 | 1.19 | ~1.27 |
| 3 | Solubility | Soluble in
1% CH3COOH |
Soluble in
1% CH3COOH |
Soluble in
1% CH3COOH |
Ash content:
The amount of ash indicates how well the demineralization process removed minerals. Table 1 shows that the ash content of the pure and synthesized chitosan were 1.19% and 1.22%, respectively, whereas the reported ash level of the commercially available chitosan was ⁓ 1.27% as can be seen from Figure 2. By comparing the parameters such as, the moisture content, ash content, and solubility, it can be concluded that pure chitosan was successfully recovered from waste shrimp shells.
FTIR Spectroscopy:
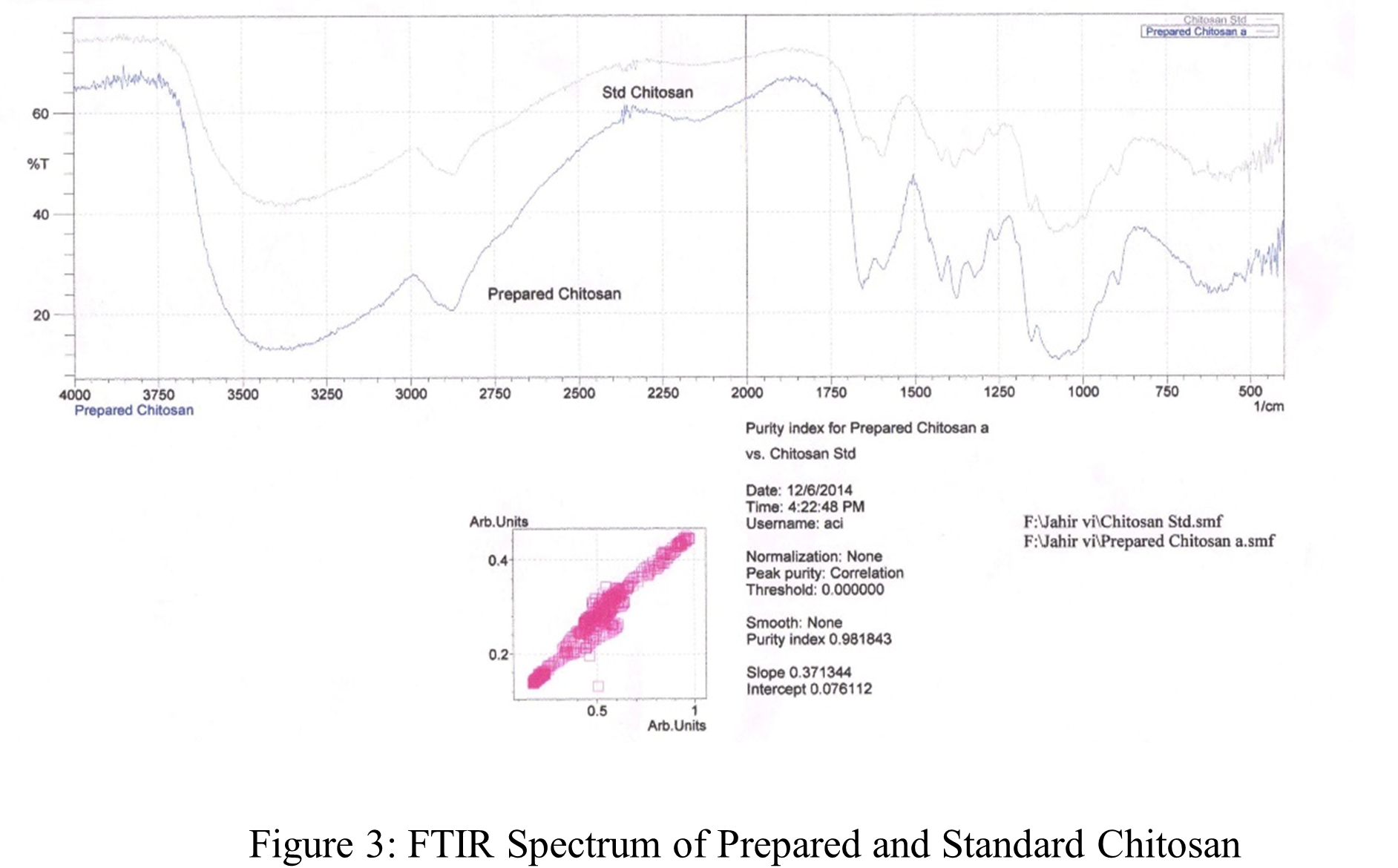
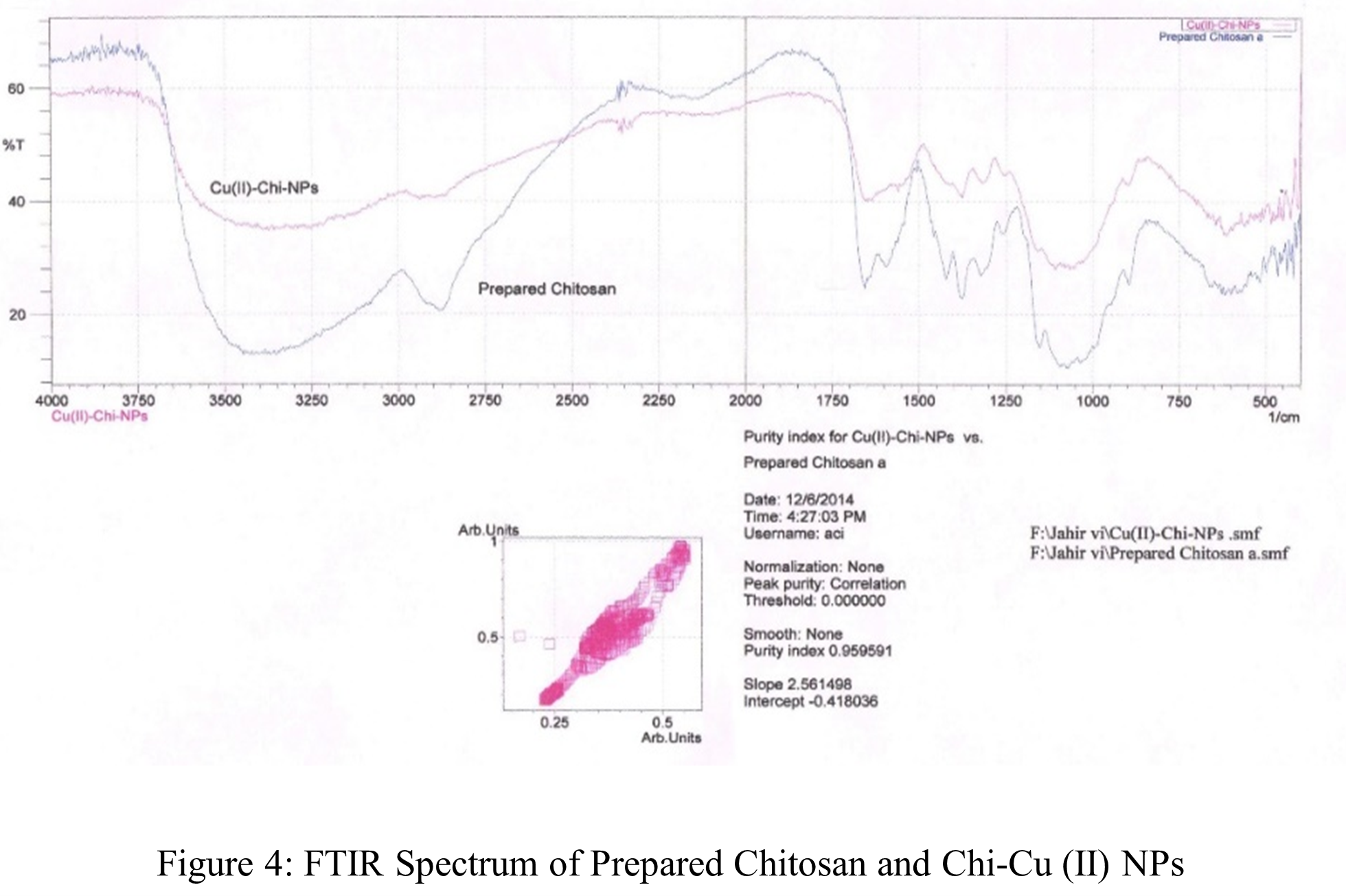
Figure 3 shows the synthesized and reference FTIR spectra of chitosan. The upper spectrum in the figure represents standard chitosan, whereas the bottom spectrum represents the synthesized chitosan. The large absorption band at 3450 – 3400 cm-1 may be the result of -OH and amine N–H symmetrical stretching vibrations, as the Figure 3 shows. The pyranose ring was shown to be responsible for a symmetric -CH2– stretching vibration that peaked at 2950 – 2800 cm-1 [79]. A peak of 1156 cm-1 is assigned to the saccharide structure. -CH3 was identified as the source of the high peak at 1350 cm-1 in the amide group [83]. The large peaks at 1021 and 1098 cm-1 revealed the presence of a C-O stretching vibration in the chitosan, whereas the peaks at 1675 and 1600 cm-1 were indicative of -C=O and -NH2 stretching in the amide I and II. The absorption bands at 1200 cm-1 were identified as the C-O-C bridges anti-symmetric stretching, while the bands at 1100 – 1020 cm-1 were identified as the skeletal vibrations associated with the C-O stretching. The results are also tabulated in Table 2. Consequently, it can be demonstrated that the spectra of synthesized and extra pure chitosan are almost the similar. This result clearly demonstrates that the chitosan was successfully separated from the remaining shrimp shell.
Table 2: FTIR studies of pure chitosan
| Wave length, (cm-1) | mode & resolution | Tentative band |
| 3450-3400 | symmetric stretching, very broad | -OH and amine N-H |
| 2950-2800 | symmetric stretching, broad | -CH2 (pyranose ring) |
| 1675 | symmetric stretching, medium | -C=O (amide I) |
| 1600 | symmetric stretching, medium | -NH (amide II) |
| 1350 | bending, sharp | -CH2 in amide group |
| 1200 | anti-symmetric stretching | C-O-C bridge |
| 1100-1020 | vibrations, broad | -C-O |
Chitosan Based Metal Nanoparticle:
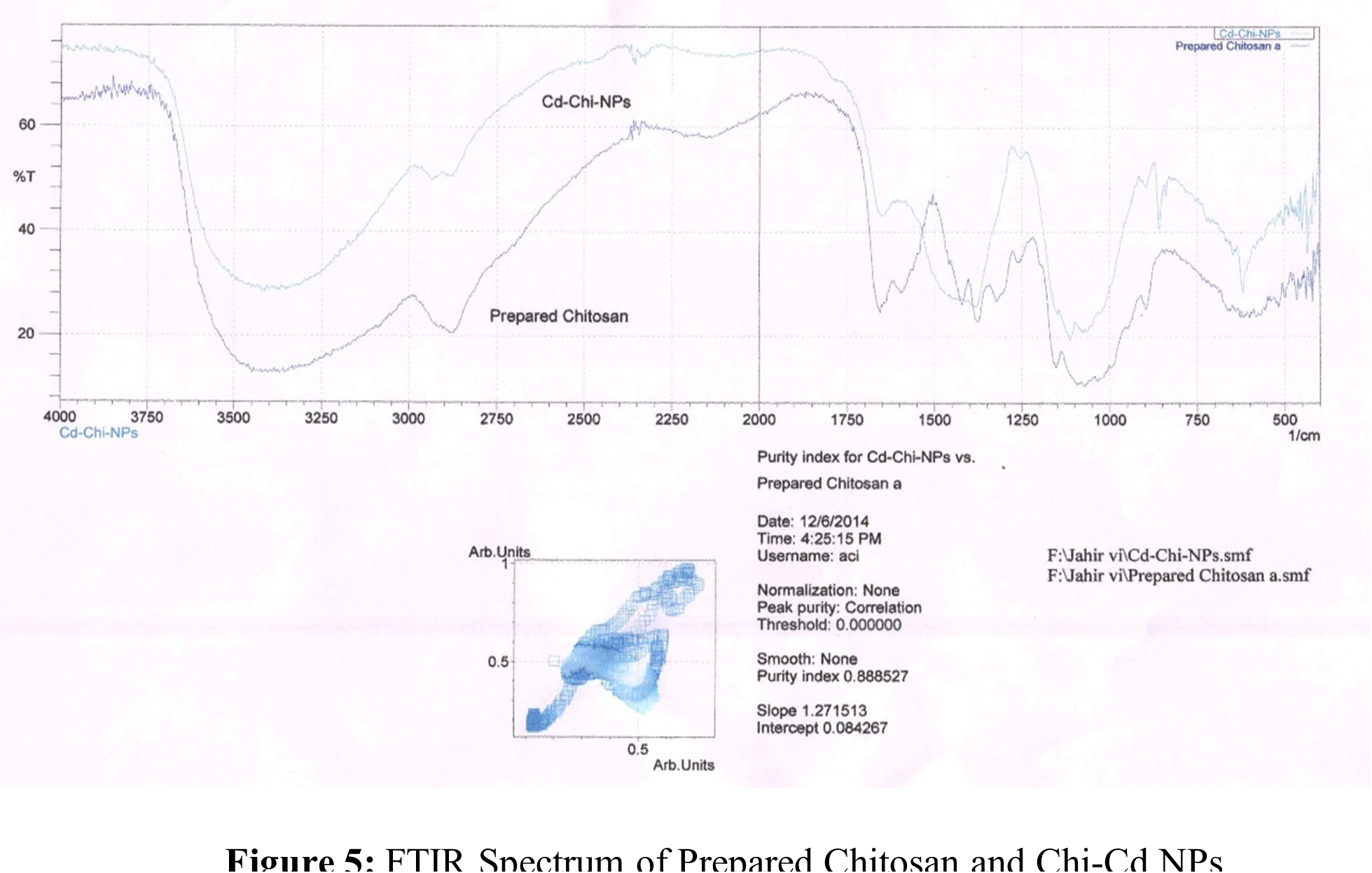
The solution cast process was adapted for producing metal nanoparticles based on chitosan. Chi-Cu(II) and Chi-Cd NPs, two distinct NPs with varying sizes and forms, were synthesized. Every one of the NPs has a distinct color based on the individual raw ingredients that were employed in the synthesis. This suggests that NPs were generated using a chemical approach. Chi-Cu(II) NPs and Chi-Cd NPs were produced using identical experimental procedures. FTIR, TGA and SEM measurements were used to analyze the produced NPs. Table 3, 4 and Figure 4, 5 display the FTIR spectra and data. The FTIR shows that, all of the NPs spectra are extremely similar to those of the chitosan that were produced. This suggests that the NPs matrices contain chitosan polymer.
Table 3: FTIR studies of Chi-Cu (II) NPs.
| Wave length, (cm-1) | mode & resolution | Tentative band |
| 3450-3400 | symmetric stretching, broad | -OH and amine N-H |
| 2950-2800 | symmetric stretching, broad | -CH2 (pyranose ring) |
| 1675 | symmetric stretching, medium | -C=O (amide I) |
| 1600 | symmetric stretching, medium | -NH (amide II) |
| 1350 | bending, sharp | -CH2 in amide group |
| 1200 | anti-symmetric stretching | C-O-C bridge |
| 1100-1020 | vibrations, broad | -C-O |
Table 4: FTIR studies of Chi-Cd NPs.
| Wave length, (cm-1) | mode & resolution | Tentative band |
| 3450-3400 | symmetric stretching, very broad | -OH and amine N-H |
| 2950-2800 | symmetric stretching, broad | -CH2 (pyranose ring) |
| 1675 | symmetric stretching, medium | -C=O (amide I) |
| 1600 | symmetric stretching, medium | -NH2 (amide II) |
| 1350 | bending, sharp | -CH2 in amide group |
| 1200 | anti-symmetric stretching | C-O-C bridge |
| 1100-1020 | vibrations, broad | -C-O |
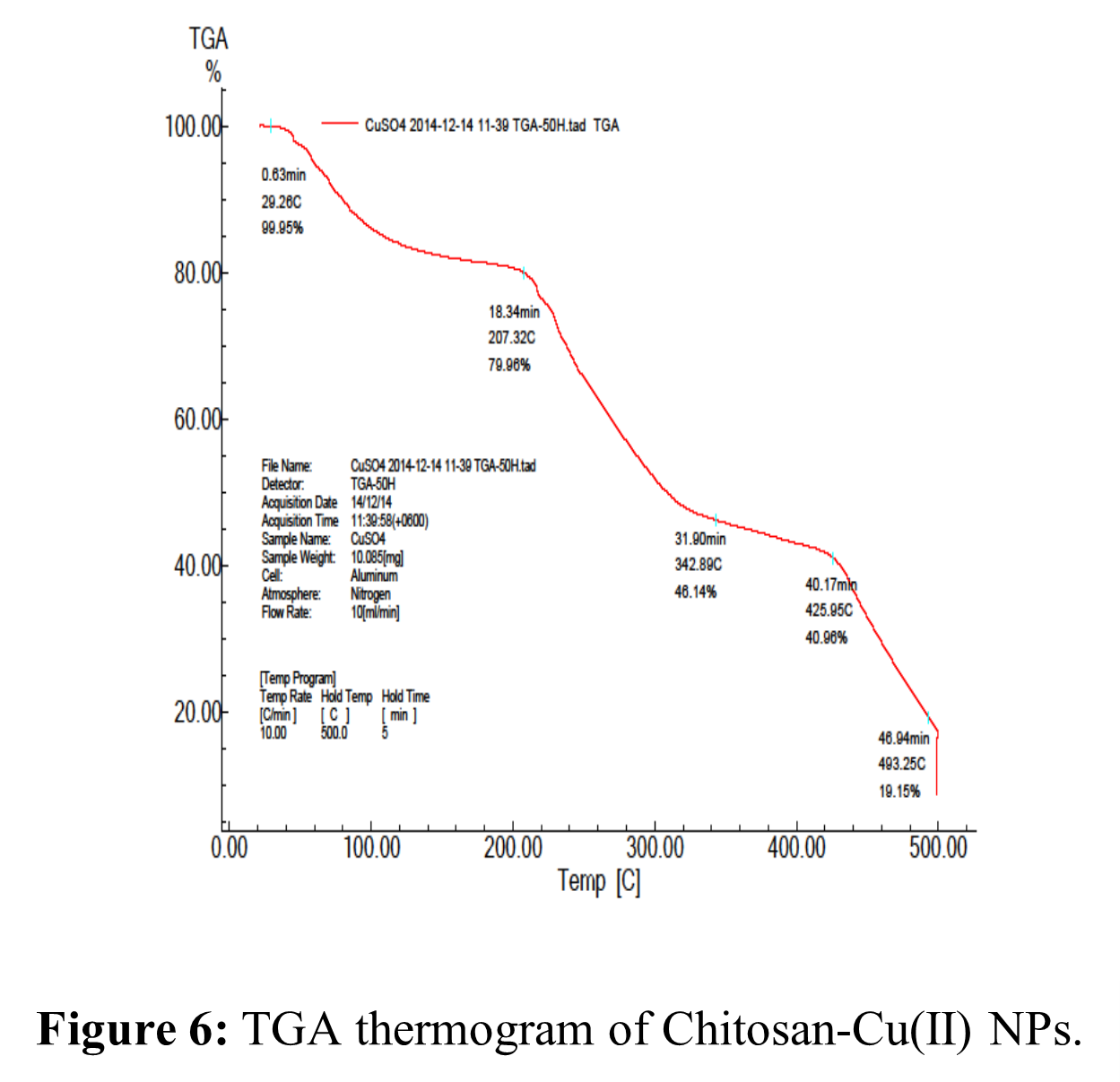
TGA of Chi-Cu(II) NPs:
Chi-Cu(II) NPs thermal details are displayed in Table 5 and Figure 6. Figure 6 shows that the Chi-Cu(II) composite Nano-material showed three successive phases of weight decrease for TG analysis. The first weight loss at 45 – 200 ºC was approximately 20.04 weight percent, which may have contributed to the crystalline water and adhering water molecules’ moisture content loss. The splitting of the ether bond in the chitosan backbone caused the second weight loss, which was approximately 39.00 wt.% in the 200 – 420 ºC range. In the third stage, the weight loss was around 21.81 weight percent in the 400 – 500 ºC range. This could be related to the glucosamine residues’ heat degradation, which results in the loss of the organic portion of the NPs [79, 84].
Table 5: TGA studies of Chi-Cu (II) NPs.
| Percentage of Decomposition (%) | Decomposition Temperature (ºC) |
| 10 | 90 |
| 20 | 210 |
| 30 | 250 |
| 40 | 280 |
| 50 | 320 |
| 60 | 420 |
| 70 | 460 |
| 80 | 495 |
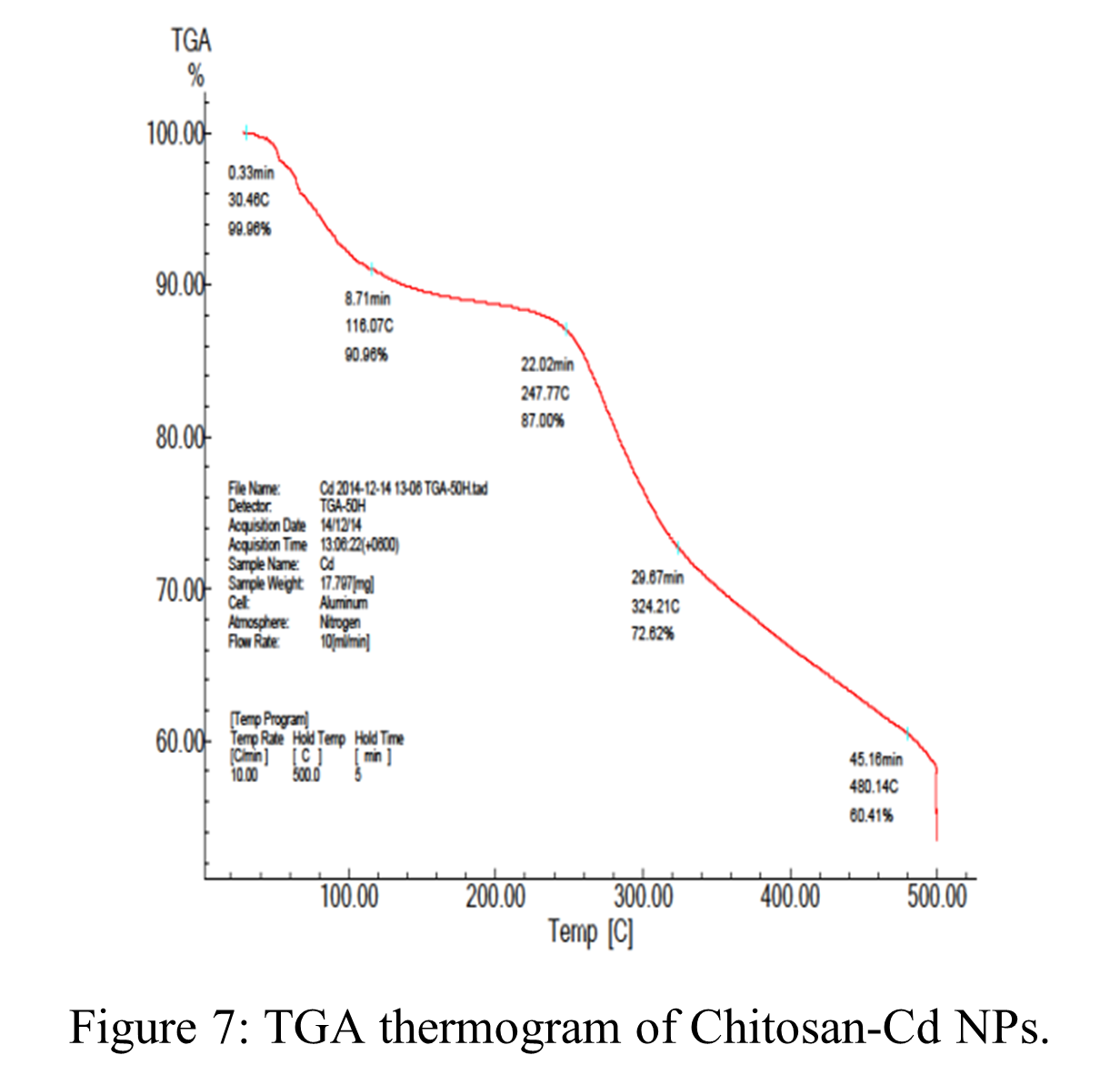
TGA of Chi-Cd NPs:
Chi-Cd NP TGA details are displayed in Table 6 and Figure 7. Three separate weight reduction phases were noted in the composite material, as shown in Figure 7. The initial weight loss at 45 – 180 ºC was almost 15% of the total weight, which may have contributed to the moisture content of crystalline water and adhering water molecules which lost on continuous decomposition. The second weight loss, which occurred between 220 and 320 ºC and was caused by the chitosan backbone’s ether linkage scission, was roughly 13.38 weight percent. The weight loss in the third stage was approximately 10.62 weight percent in the 320 – 500 ºC range, which could be related to the glucosamine residues’ heat degradation, which implies the NPs’ organic components were being lost [79, 84]. However, the problems associated with the weight loss machine, further experiments could not be continued onwards.
Table 6: TGA studies of Chi-Cd NPs.
| Percentage of Decomposition (%) | Decomposition Temperature (ºC) |
| 10 | 110 |
| 20 | 280 |
| 30 | 340 |
| 40 | 500 |
SEM analysis of Chi-Cu (II) NPs:
Chi-Cu (II) NPs SEM image was obtained at X5,000 and X25,000 magnifications. The specific NPs could not be distinguished in this case. However, based on Figure 8, it can be inferred that Chi-Cu (II) NPs possessed morphologies that were similar to flakes in shape and had particle sizes that were within 100 nm.
SEM analysis of Chi-Cd NPs:
Chi-Cd NPs were imaged using a SEM at magnifications of X10,000 and X25,000. The nanoparticle images, which are displayed in Figure 9, demonstrated that the majority of Chi-Cd NP morphologies possessed spherical shapes and particle size ranging from 1 to 5 nm.
Antibacterial Analysis:
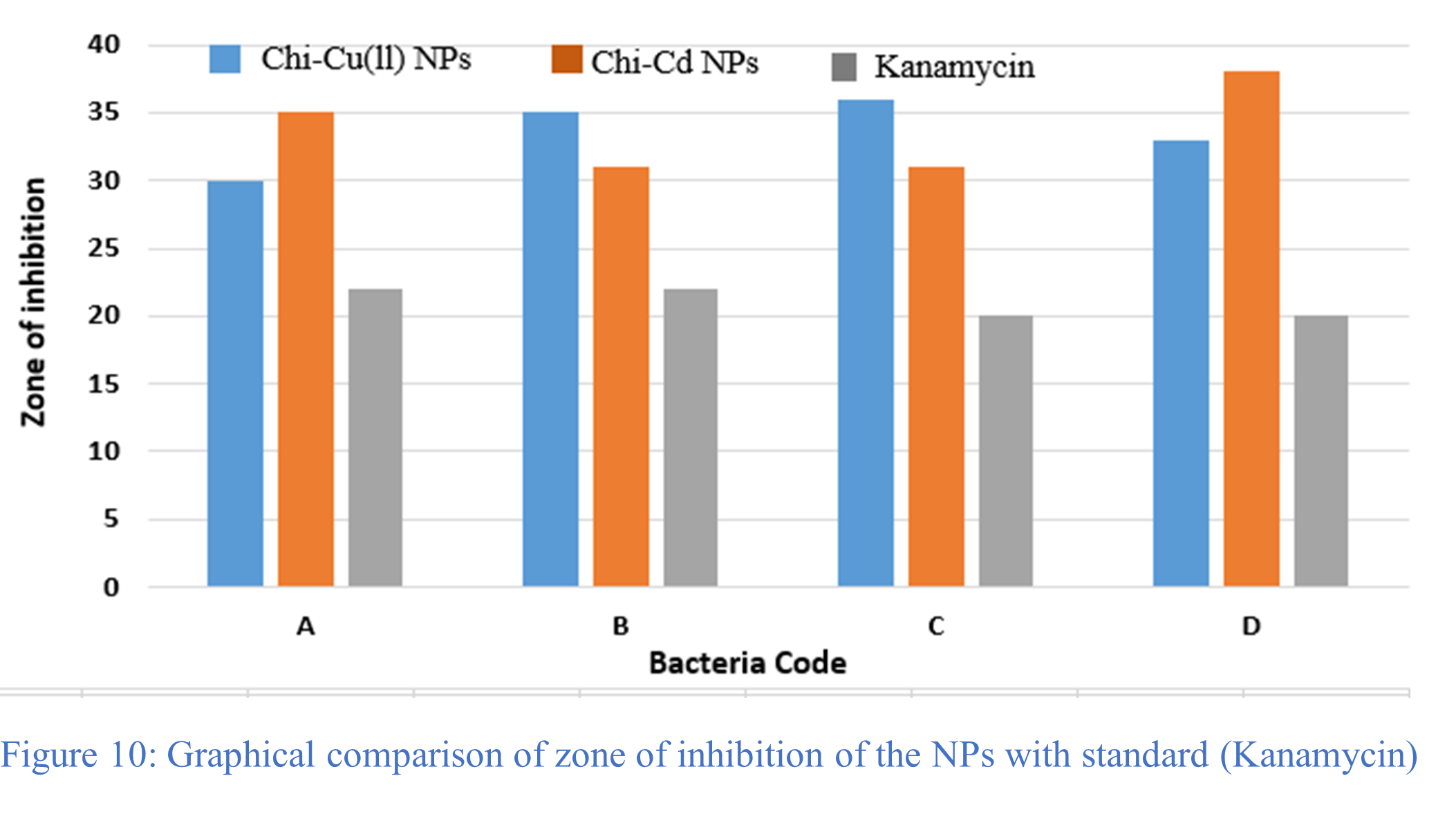
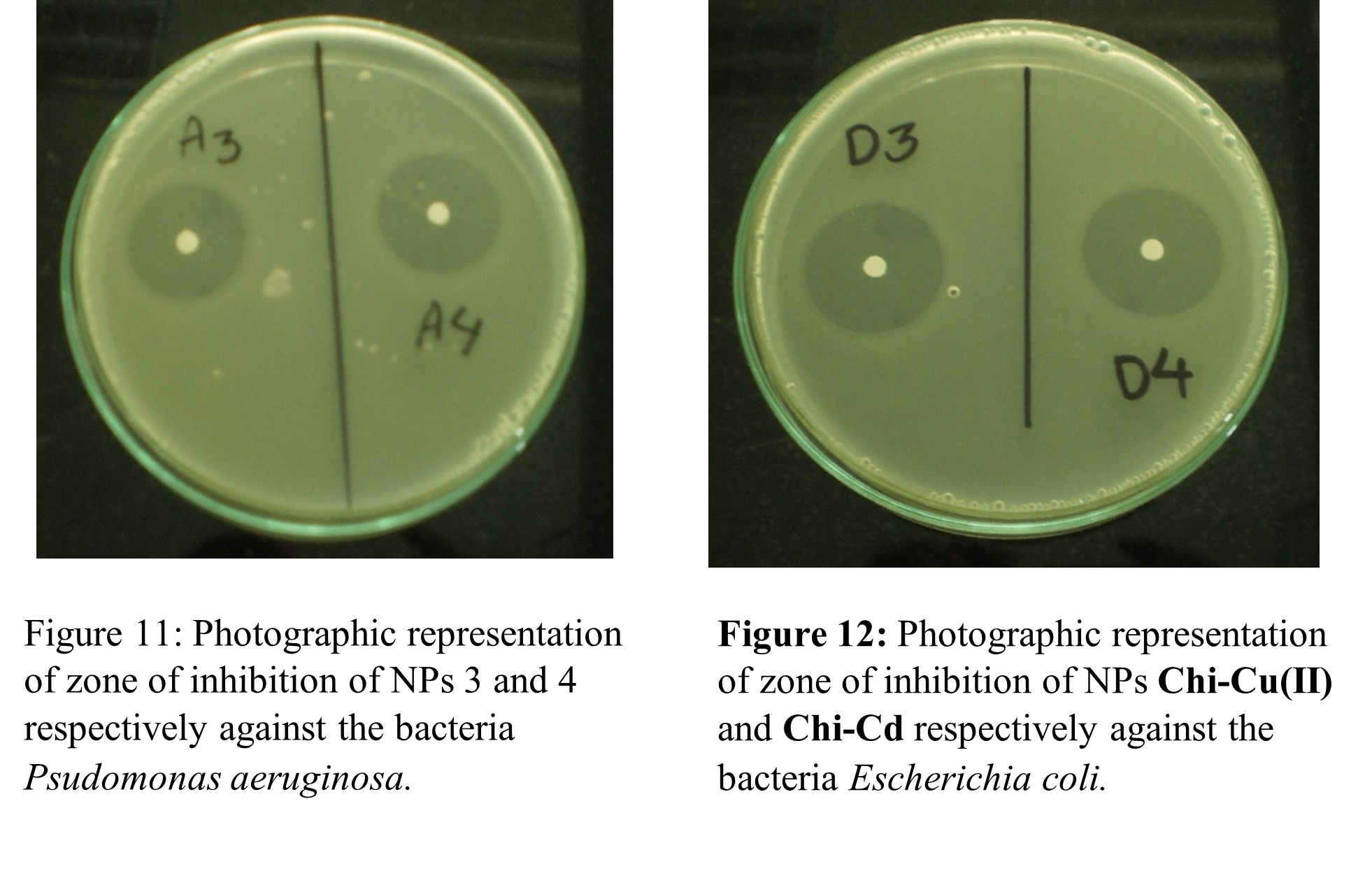
The antibacterial efficacy of the targeted chitosan-metal NPs against gram-negative bacteria, which included Pseudomonas aeruginosa, Salmonella bovismorbificans, Salmonella typhi, and Escherichia coli, were investigated biologically. The target compounds’ antibacterial activity in terms of zone of inhibition on agar disk diffusion plates are shown in Figures 11 and 12. Table 7 provides the findings of diameters of zone of inhibition in mm (millimeters) of studied NPs and standard antibiotic, kanamycin. The zone of inhibition for the kanamycin and for the gram-negative bacteria Pseudomonas aeruginosa, Salmonella bovismorbificans, Salmonella typhi, and Escherichia coli were found to be 22, 22, 20, and 20 mm, respectively. Figure 10 illustrates a graphical comparison of the zone of inhibition for the standard and chitosan-metal nanoparticles against the bacteria. Therefore, this antimicrobial activity analysis shows that all of the produced NPs were found to incredibly active against bacteria. The potency of each NP is significantly higher than that of the standard antibiotic, kanamycin.
Table 7: Results of the antibacterial activity of the different NPs.
| Bacteria code | Name of the bacteria | Diameter of zone of inhibition of bacteria in different NPs* (mm) | ||
| Chi-Cu(II) NPs 30 µg/disk | Chi-Cd NPs 30 µg/disk | Kanamycin 30 µg/disk | ||
| A | Psudomonas aeruginosa (-ve) | 30 | 35 | 22 |
| B | Salmonella bovis morbificans(-ve) | 35 | 31 | 22 |
| C | Salmonella typhi (-ve) | 36 | 31 | 20 |
| D | Escherichia coli (-ve) | 33 | 38 | 20 |
CONCLUSION
We have successfully prepared chitosan from shrimp shell and synthesized chitosan-metal NPs. The prepared chitosan’s properties nicely aligned with that of standard chitosan and found to be attained better features among the studied parameters than available commercial chitosan. The procedures developed for the preparation of chitosan nanoparticles are very simple and easy to perform. In particular, Chi-Cd NPs preparation method was utilized for the first time in this research. Both Chi-Cu(II) NPs and Chi-Cd NPs showed greater antibacterial activity than the standard kanamycin. The procedure of synthesis of Cd and Cu (II) nanoparticles explored in this work would be environmentally friendly, safe, and fast, which could be commercially viable in a long run. Moreover, this method can also be implemented on industrial scale in order to synthesize NPs and furthermore, prepared NPs can also be used as an effective antibacterial agent.
CONFLICT OF INTEREST
The authors have declared that they do not have any conflict of interest regarding publication of this research.
ACKNOWLEDGEMENTS
Authors gratefully acknowledge the UGC Bangladesh for research grants; Department of Chemistry, Khulna University of Engineering & Technology, Bangladesh; University of Dhaka, Bangladesh; ACI Pharmaceuticals Ltd. Bangladesh and Department of Fisheries, Fish Inspection & Quality Control microbiological laboratory in Khulna, Bangladesh for the facilities provided to conduct the research.
REFERENCES
- N. Morin-Crini, E. Lichtfouse, G. Torri, G. Crini, Fundamentals and applications of chitosan, Sustainable agriculture reviews 35: chitin and chitosan: history, fundamentals and innovations, (2019) 49-123.
- I. Younes, M. Rinaudo, Chitin and chitosan preparation from marine sources. Structure, properties and applications, Marine drugs, 13 (2015) 1133-1174.
- A. Varma, S. Deshpande, J. Kennedy, Metal complexation by chitosan and its derivatives: a review, Carbohydrate polymers, 55 (2004) 77-93.
- S. Hudson, C. Smith, Polysaccharides: chitin and chitosan: chemistry and technology of their use as structural materials, Biopolymers from renewable resources, Springer1998, pp. 96-118.
- T.D. Rathke, S.M. Hudson, Review of chitin and chitosan as fiber and film formers, Journal of Macromolecular Science, Part C: Polymer Reviews, 34 (1994) 375-437.
- V.S. Yeul, S.S. Rayalu, Unprecedented chitin and chitosan: A chemical overview, Journal of Polymers and the Environment, 21 (2013) 606-614.
- T.-C. Chou, E. Fu, C.-J. Wu, J.-H. Yeh, Chitosan enhances platelet adhesion and aggregation, Biochemical and Biophysical Research Communications, 302 (2003) 480-483.
- P. Schulz, M. Rodriguez, L. Del Blanco, M. Pistonesi, E. Agullo, Emulsification properties of chitosan, Colloid and Polymer Science, 276 (1998) 1159-1165.
- T. Wu, S. Zivanovic, F.A. Draughon, W.S. Conway, C.E. Sams, Physicochemical properties and bioactivity of fungal chitin and chitosan, Journal of agricultural and food chemistry, 53 (2005) 3888-3894.
- S. Kumari, S.H.K. Annamareddy, S. Abanti, P.K. Rath, Physicochemical properties and characterization of chitosan synthesized from fish scales, crab and shrimp shells, International journal of biological macromolecules, 104 (2017) 1697-1705.
- J.-K. Hwang, H.-H. Shin, Rheological properties of chitosan solutions, Korea-Australia Rheology Journal, 12 (2000) 175-179.
- [12] R.R. Klossner, H.A. Queen, A.J. Coughlin, W.E. Krause, Correlation of chitosan’s rheological properties and its ability to electrospin, Biomacromolecules, 9 (2008) 2947-2953.
- W. Wang, Q. Meng, Q. Li, J. Liu, M. Zhou, Z. Jin, K. Zhao, Chitosan derivatives and their application in biomedicine, International journal of molecular sciences, 21 (2020) 487.
- R. Muzzarelli, C. Muzzarelli, Chitosan chemistry: relevance to the biomedical sciences, Polysaccharides I: structure, characterization and use, (2005) 151-209.
- F. Bahavarnia, M. Hasanzadeh, P. Bahavarnia, N. Shadjou, Advancements in application of chitosan and cyclodextrins in biomedicine and pharmaceutics: recent progress and future trends, RSC advances, 14 (2024) 13384-13412.
- G. Kravanja, M. Primožič, Ž. Knez, M. Leitgeb, Chitosan-based (Nano) materials for novel biomedical applications, Molecules, 24 (2019) 1960.
- L. Ilium, Chitosan and its use as a pharmaceutical excipient, Pharmaceutical research, 15 (1998) 1326-1331.
- Z. Shariatinia, Pharmaceutical applications of chitosan, Advances in colloid and interface science, 263 (2019) 131-194.
- M.R. Kumar, R.A. Muzzarelli, C. Muzzarelli, H. Sashiwa, A. Domb, Chitosan chemistry and pharmaceutical perspectives, Chemical reviews, 104 (2004) 6017-6084.
- C. Verma, M. Quraishi, Chelation capability of chitosan and chitosan derivatives: Recent developments in sustainable corrosion inhibition and metal decontamination applications, Current Research in Green and Sustainable Chemistry, 4 (2021) 100184.
- R.A. Muzzarelli, M. De Vincenzi, Chitosans as dietary food additives, Applications of Chitan and Chitosan, CRC Press2020, pp. 115-127.
- E. Agulló, M.S. Rodríguez, V. Ramos, L. Albertengo, Present and future role of chitin and chitosan in food, Macromolecular bioscience, 3 (2003) 521-530.
- I. Hamed, F. Özogul, J.M. Regenstein, Industrial applications of crustacean by-products (chitin, chitosan, and chitooligosaccharides): A review, Trends in food science & technology, 48 (2016) 40-50.
- A. Rafique, K.M. Zia, M. Zuber, S. Tabasum, S. Rehman, Chitosan functionalized poly (vinyl alcohol) for prospects biomedical and industrial applications: A review, International journal of biological macromolecules, 87 (2016) 141-154.
- V. Chavasit, J.A. Torres, Chitosan‐Poly (acrylic acid): Mechanism of complex formation and potential industrial applications, Biotechnology progress, 6 (1990) 2-6.
- S. Rodrigues, M. Dionísio, C. Remunan Lopez, A. Grenha, Biocompatibility of chitosan carriers with application in drug delivery, Journal of functional biomaterials, 3 (2012) 615-641.
- T. Kean, M. Thanou, Biodegradation, biodistribution and toxicity of chitosan, Advanced drug delivery reviews, 62 (2010) 3-11.
- A. Schneider, L. Richert, G. Francius, J.-C. Voegel, C. Picart, Elasticity, biodegradability and cell adhesive properties of chitosan/hyaluronan multilayer films, Biomedical materials, 2 (2007) S45.
- N.A. Dzung, N.T.N. Hà, D.T.H. Van, N.T.L. Phuong, N.T.N. Quynh, D.M. Hiep, Chitosan nanoparticle as a novel delivery system for A/H1n1 influenza vaccine: safe property and immunogenicity in mice, International Journal of Biotechnology and Bioengineering, 5 (2011) 915-922.
- S. Moratti, J. Cabral, Antibacterial properties of chitosan, Chitosan Based Biomaterials Volume 1, Elsevier2017, pp. 31-44.
- S. Malik, K. Muhammad, Y. Waheed, Nanotechnology: A revolution in modern industry, Molecules, 28 (2023) 661.
- L. Pokrajac, A. Abbas, W. Chrzanowski, G.M. Dias, B.J. Eggleton, S. Maguire, E. Maine, T. Malloy, J. Nathwani, L. Nazar, Nanotechnology for a sustainable future: Addressing global challenges with the international network4sustainable nanotechnology, ACS Publications, 2021.
- A.G. Ingale, A. Chaudhari, Biogenic synthesis of nanoparticles and potential applications: an eco-friendly approach, J Nanomed Nanotechol, 4 (2013) 1-7.
- S.F. Ahmed, M. Mofijur, N. Rafa, A.T. Chowdhury, S. Chowdhury, M. Nahrin, A.S. Islam, H.C. Ong, Green approaches in synthesising nanomaterials for environmental nanobioremediation: Technological advancements, applications, benefits and challenges, Environmental Research, 204 (2022) 111967.
- M. Fakruddin, Z. Hossain, H. Afroz, Prospects and applications of nanobiotechnology: a medical perspective, Journal of nanobiotechnology, 10 (2012) 1-8.
- M.C. Roco, Nanotechnology: convergence with modern biology and medicine, Current opinion in biotechnology, 14 (2003) 337-346.
- A. Boroumand Moghaddam, F. Namvar, M. Moniri, P. Md. Tahir, S. Azizi, R. Mohamad, Nanoparticles biosynthesized by fungi and yeast: a review of their preparation, properties, and medical applications, Molecules, 20 (2015) 16540-16565.
- P. Singh, A. Garg, S. Pandit, V. Mokkapati, I. Mijakovic, Antimicrobial effects of biogenic nanoparticles, Nanomaterials, 8 (2018) 1009.
- W. Gao, J.C. Lai, S.W. Leung, Functional enhancement of chitosan and nanoparticles in cell culture, tissue engineering, and pharmaceutical applications, Frontiers in physiology, 3 (2012) 321.
- S. Bhatia, S. Bhatia, Chitosan based nanomaterials and its applications, Systems for Drug Delivery: Safety, Animal, and Microbial Polysaccharides, (2016) 55-117.
- S. Rashki, K. Asgarpour, H. Tarrahimofrad, M. Hashemipour, M.S. Ebrahimi, H. Fathizadeh, A. Khorshidi, H. Khan, Z. Marzhoseyni, M. Salavati-Niasari, Chitosan-based nanoparticles against bacterial infections, Carbohydrate polymers, 251 (2021) 117108.
- W. Tang, J. Wang, H. Hou, Y. Li, J. Wang, J. Fu, L. Lu, D. Gao, Z. Liu, F. Zhao, Application of chitosan and its derivatives in medical materials, International Journal of Biological Macromolecules, (2023) 124398.
- J.A.C. Pérez, J.E. Sosa-Hernández, S.M. Hussain, M. Bilal, R. Parra-Saldivar, H.M. Iqbal, Bioinspired biomaterials and enzyme-based biosensors for point-of-care applications with reference to cancer and bio-imaging, Biocatalysis and agricultural biotechnology, 17 (2019) 168-176.
- A.M. Abdelaziz, S. Dacrory, A.H. Hashem, M.S. Attia, M. Hasanin, H.M. Fouda, S. Kamel, H. ElSaied, Protective role of zinc oxide nanoparticles based hydrogel against wilt disease of pepper plant, Biocatalysis and Agricultural Biotechnology, 35 (2021) 102083.
- H.E. Abdelmoneim, M.A. Wassel, A.S. Elfeky, S.H. Bendary, M.A. Awad, S.S. Salem, S.A. Mahmoud, Multiple applications of CdS/TiO 2 nanocomposites synthesized via microwave-assisted sol–gel, Journal of Cluster Science, (2022) 1-10.
- E. Saied, A.M. Eid, S.E.-D. Hassan, S.S. Salem, A.A. Radwan, M. Halawa, F.M. Saleh, H.A. Saad, E.M. Saied, A. Fouda, The catalytic activity of biosynthesized magnesium oxide nanoparticles (MgO-NPs) for inhibiting the growth of pathogenic microbes, tanning effluent treatment, and chromium ion removal, Catalysts, 11 (2021) 821.
- J. Tanwar, S. Das, Z. Fatima, S. Hameed, Multidrug resistance: an emerging crisis, Interdisciplinary perspectives on infectious diseases, 2014 (2014).
- Z.-Q. Xu, M.T. Flavin, J. Flavin, Combating multidrug-resistant Gram-negative bacterial infections, Expert opinion on investigational drugs, 23 (2014) 163-182.
- R. Huwaitat, A.P. McCloskey, B.F. Gilmore, G. Laverty, Potential strategies for the eradication of multidrug-resistant Gram-negative bacterial infections, Future microbiology, 11 (2016) 955-972.
- M.-j. Medina, H. Legido-Quigley, L.Y. Hsu, Antimicrobial resistance in one health, Global Health Security: Recognizing Vulnerabilities, Creating Opportunities, (2020) 209-229.
- F. Bongomin, S. Gago, R.O. Oladele, D.W. Denning, Global and multi-national prevalence of fungal diseases—estimate precision, Journal of fungi, 3 (2017) 57.
- M.A. Pfaller, Antifungal drug resistance: mechanisms, epidemiology, and consequences for treatment, The American journal of medicine, 125 (2012) S3-S13.
- Y. Ohya, M. Shiratani, H. Kobayashi, T. Ouchi, Release behavior of 5-fluorouracil from chitosan-gel nanospheres immobilizing 5-fluorouracil coated with polysaccharides and their cell specific cytotoxicity, Journal of Macromolecular Science—Pure and Applied Chemistry, 31 (1994) 629-642.
- H. Liu, B. Chen, Z. Mao, C. Gao, Chitosan nanoparticles for loading of toothpaste actives and adhesion on tooth analogs, Journal of Applied Polymer Science, 106 (2007) 4248-4256.
- A. Grenha, M.E. Gomes, M. Rodrigues, V.E. Santo, J.F. Mano, N.M. Neves, R.L. Reis, Development of new chitosan/carrageenan nanoparticles for drug delivery applications, Journal of Biomedical Materials Research Part A: An Official Journal of The Society for Biomaterials, The Japanese Society for Biomaterials, and The Australian Society for Biomaterials and the Korean Society for Biomaterials, 92 (2010) 1265-1272.
- B. Sarmento, S. Martins, A. Ribeiro, F. Veiga, R. Neufeld, D. Ferreira, Development and comparison of different nanoparticulate polyelectrolyte complexes as insulin carriers, International Journal of Peptide Research and Therapeutics, 12 (2006) 131-138.
- J. Balcerzak, M. Kucharska, B. Gruchała, Preparation of micro and nanostructures of chitosan by ultrasonic coalescence of w/o emulsions, Progress on Chemistry and Application of Chitin and its Derivatives, 18 (2013) 13-20.
- K. Tahara, T. Sakai, H. Yamamoto, H. Takeuchi, N. Hirashima, Y. Kawashima, Improvements in transfection efficiency with chitosan modified poly (DL-lactide-co-glycolide) nanospheres prepared by the emulsion solvent diffusion method, for gene delivery, Chemical and Pharmaceutical Bulletin, 59 (2011) 298-301.
- M.S. Orellano, G.S. Longo, C. Porporatto, N.M. Correa, R.D. Falcone, Role of micellar interface in the synthesis of chitosan nanoparticles formulated by reverse micellar method, Colloids and Surfaces A: Physicochemical and Engineering Aspects, 599 (2020) 124876.
- M.A. Mosa, S.E. El-Abeid, Chitosan-Loaded Copper Oxide Nanoparticles: A Promising Antifungal Nanocomposite against Fusarium Wilt Disease of Tomato Plants, Sustainability, 15 (2023) 14295.
- A. Naz, S. Arun, S.S. Narvi, M.S. Alam, A. Singh, P. Bhartiya, P. Dutta, Cu (II)-carboxymethyl chitosan-silane schiff base complex grafted on nano silica: Structural evolution, antibacterial performance and dye degradation ability, International journal of biological macromolecules, 110 (2018) 215-226.
- N.-Y. Nguyen, B.N. An, M.-V. Le, H.A. Hoang, Antibacterial activity of copper nanoparticles-chitosan composite against Vibrio parahaemolyticus, Biocontrol Science, 25 (2020) 159-165.
- N. Cioffi, L. Torsi, N. Ditaranto, G. Tantillo, L. Ghibelli, L. Sabbatini, T. Bleve-Zacheo, M. D’Alessio, P.G. Zambonin, E. Traversa, Copper nanoparticle/polymer composites with antifungal and bacteriostatic properties, Chemistry of Materials, 17 (2005) 5255-5262.
- M. Taner, N. Sayar, I.G. Yulug, S. Suzer, Synthesis, characterization and antibacterial investigation of silver–copper nanoalloys, Journal of Materials Chemistry, 21 (2011) 13150-13154.
- M.C. Fung, D.L. Bowen, Silver products for medical indications: risk-benefit assessment, Journal of toxicology: Clinical toxicology, 34 (1996) 119-126.
- M.H. Habibi, R. Kamrani, R. Mokhtari, Fabrication and characterization of copper nanoparticles using thermal reduction: the effect of nonionic surfactants on size and yield of nanoparticles, Microchimica Acta, 171 (2010) 91-95.
- A.A. Ponce, K.J. Klabunde, Chemical and catalytic activity of copper nanoparticles prepared via metal vapor synthesis, Journal of Molecular Catalysis A: Chemical, 225 (2005) 1-6.
- A. Khan, A. Rashid, R. Younas, R. Chong, A chemical reduction approach to the synthesis of copper nanoparticles, International Nano Letters, 6 (2016) 21-26.
- M.R. Abdullah, N.H. Harun, S.N. Ibrahim, A.A. Wahab, Thin film coating of copper nanoparticles with DC magnetron sputtering via physical vapor deposition, AIP Conference Proceedings, AIP Publishing, 2019.
- H.-C. Tu, H.-W. Hsieh, K.-S. Ho, A facile way to prepare tiny Cu nanoparticles from micro-emulsion micelles of Cu (II) di-n-dodecylbenzene sulfonate, Colloids and Surfaces A: Physicochemical and Engineering Aspects, (2024) 133378.
- R. Tilaki, A. Iraji Zad, S. Mahdavi, Size, composition and optical properties of copper nanoparticles prepared by laser ablation in liquids, Applied Physics A, 88 (2007) 415-419.
- R.R. Prabhu, M.A. Khadar, Characterization of chemically synthesized CdS nanoparticles, Pramana, 65 (2005) 801-807.
- N. Hullavarad, S. Hullavarad, P. Karulkar, Cadmium sulphide (CdS) nanotechnology: synthesis and applications, Journal of nanoscience and nanotechnology, 8 (2008) 3272-3299.
- H.K. No, E.Y. Hur, Control of foam formation by antifoam during demineralization of crustacean shell in preparation of chitin, Journal of Agricultural and Food Chemistry, 46 (1998) 3844-3846.
- A. Percot, C. Viton, A. Domard, Characterization of shrimp shell deproteinization, Biomacromolecules, 4 (2003) 1380-1385.
- M. Seenuvasan, G. Sarojini, M. Dineshkumar, Recovery of chitosan from natural biotic waste, Current developments in biotechnology and bioengineering, Elsevier2020, pp. 115-133.
- S.S. Nielsen, Determination of moisture content, Food analysis laboratory manual, (2010) 17-27.
- B.P. Ismail, Ash content determination, Food analysis laboratory manual, (2017) 117-119.
- A. Pawlak, M. Mucha, Thermogravimetric and FTIR studies of chitosan blends, Thermochimica acta, 396 (2003) 153-166.
- J. Brugnerotto, J. Lizardi, F.M. Goycoolea, W. Argüelles-Monal, J. Desbrières, M. Rinaudo, An infrared investigation in relation with chitin and chitosan characterization, Polymer, 42 (2001) 3569-3580.
- M. Zaidan, A. Noor Rain, A. Badrul, A. Adlin, A. Norazah, I. Zakiah, In vitro screening of five local medicinal plants for antibacterial activity using disc diffusion method, Trop biomed, 22 (2005) 165-170.
- M. Krishnan, D.K. Dey, C. Sharma, S.C. Kang, Antibacterial activity of Weissella confusa by disc diffusion method, ||| Bangladesh Journal of Pharmacology, 14 (2019) 117-122.
- H. Zheng, Y. Du, J. Yu, R. Huang, L. Zhang, Preparation and characterization of chitosan/poly (vinyl alcohol) blend fibers, Journal of Applied Polymer Science, 80 (2001) 2558-2565.
- C.-H. Chen, F.-Y. Wang, C.-F. Mao, W.-T. Liao, C.-D. Hsieh, Studies of chitosan: II. Preparation and characterization of chitosan/poly (vinyl alcohol)/gelatin ternary blend films, International Journal of Biological Macromolecules, 43 (2008) 37-42.