Use of Rice Bran as Ruminant Feed in Indonesia
- Urip Rosani
- Iman Hernaman
- Rahmat Hidayat
- Darmawan Hidayat
- 489-504
- Feb 24, 2024
- Education
Use of Rice Bran as Ruminant Feed in Indonesia
Urip Rosani*1, Iman Hernaman1, Rahmat Hidayat1, Darmawan Hidayat2
1Department of Animal Nutrition and Feed Technology, Faculty of Animal Husbandry, Universitas Padjadjaran Sumedang 45363, West Java, Indonesia
2Department of Electrical Engineering, Faculty of Mathematics and Natural Sciences, Universitas Padjadjaran, Sumedang 45363, West Java, Indonesia
*Corresponding Author
DOI: https://doi.org/10.51244/IJRSI.2024.1101036
Received: 10 January 2024; Revised: 22 January 2024; Accepted: 27 January 2024; Published: 24 February 2024
ABSTRACT
The purpose of this study is to determine the potential of rice bran and rice husks as feed and their impact on ruminants, as well as how to detect rice husk contamination in rice bran. Rice bran has great potential as a feed ingredient. In addition to containing nutrients that are quite high (crude protein 8–12%), it is also easily accessible to farmers because almost every region of Indonesia produces rice bran in fairly abundant quantities. The use of rice bran as animal feed has long been widely used by some farmers in Indonesia. The need for rice bran as animal feed is not in accordance with its availability, which causes these feed ingredients to have high prices, especially in the dry season. Even today, the price of rice bran is close to the price of corn. The high cost and scarcity of rice bran cause massive counterfeiting, especially with the addition of rice husks. Even though rice husks have 25–30% lignin and 15-20% silica, which, if used in the long term, will cause livestock productivity to decrease, cause reproductive failure, decrease health quality, and cause death. Rice bran is a very potential feed ingredient for ruminants because it contains nutrients that are in accordance with their needs. The high need for rice bran is not in line with its productivity, so it is mixed with many other materials, especially rice husks. Mixing with other materials can be determined by their physical and chemical properties. Rice husks are an alternative energy source for ruminants. To improve the digestibility of rice husks through physical, chemical, and biological treatment. Recommendations for using rice husks as a feed ingredient have not been widely reported, so future research is needed.
Keywords: Rice Bran, Rice Husk, Ruminant, Feed, Contamination
INTRODUCTION
The potential need for rice bran is high, but still less than its needs, causing the price of rice bran to be high. Currently, the price of rice bran is almost equal to the price of corn, which is around Rp. 4,000. This condition is the cause of massive adulteration of rice bran by mixing it with other materials that have physical characteristics almost the same as the original raw materials to be faked.
Adulteration is the process of adding contamination to pure materials. In the feed industry, adulterated or contaminated feed can be considered unsafe and unhealthy for livestock. Feed contamination is categorized as one cause of decreased livestock productivity. The materials that have been used as counterfeiting materials for rice bran are milled rice husks, peanut shell flour, corn cob flour, and sawdust. Among these counterfeiting materials, rice husks are often added. This is because the potential of rice husks is very high, they have a high degree of similarity with rice bran, and in general, they have no price.
Rice husks are the result of rice milling and contain about 20% of dry grain. This amount has great potential if it can be used as feed. But unfortunately, until now, it has not been used as a feed ingredient because it contains high levels of lignin and silica, so it can interfere with livestock productivity and health if used for a long time. In cases found in the field with dairy cows in the working area of the Bayong Bong KUD, the use of rice bran mixed with high-milled rice husks (above 50%) in dairy cows causes milk production, health, and reproductive systems to be disrupted. This will disturb the level of trust of farmers in feed producers, and feed producers are also harmed because the quality of rice bran does not match the price paid to rice bran suppliers. In order to cheat and prove the existence of counterfeiting rice bran by husks, accurate and irrefutable evidence is needed. Therefore, a method of testing the quality of rice bran is needed.
The effect of rice bran contaminated with rice husks greatly affects the fermentability, digestibility, and performance of sheep. This is because rice husks contain lignocellulose and silica complexes that are difficult for microbes to digest. The higher the rice husk content in rice bran, the lower the amount of nutrient intake for livestock because it cannot be digested and utilized by livestock. Livestock nutrient needs are not met, so it will interfere with livestock performance.
Rice husks have 25-30% lignin and 15-20% silica [2] which if used in the long term will cause livestock productivity to decrease and cause reproductive failure, decrease the quality of health and cause death.
Lignin is a complex aromatic polymer in the cell wall, which increases with the development of plant maturity. It consists of phenylpropane units [3], Filling the gap between cellulose and hemicellulose is similar to resin [4]. In addition, it is known that lignin content provides structural strength and rigidity to plant cell walls. In the rumen, lignin restricts microbial enzymatic access to lignocellulose, which affects the digestibility of fibre [5] and greatly interferes with its utilization rate. As the lignin content decreases, the digestibility rate of dry matter increases significantly [6] due to the nature of lignin inhibits the activity of rumen microbial enzymes [7]. With low digestibility of fibre, it will have an impact on fermentation products in the form of VFA which decreases as well [8], [9] where VFA is used as an energy source for ruminants both for toys and production [10]. Thus, the presence of high lignin will have an impact on the performance of ruminants [11].
Rice husks can produce ash with high silica content. Rice husks, when burned will produce about 20% husk ash. The ash contains between 92-95% silica, with high porosity, light weight and a wide external surface [12]. Silica particles affect nutrient absorption [13] and gastrointestinal conditions, therefore can exert toxic effects [14].
Silica lowers palatability and prevents colonization of rumen microorganisms [15], thus interfering with the degradability of feed. Every 1% increase in silica content of the material will decrease digestibility by 2 – 3% in ruminants [16]. Excess intake of silica causes urolithiasis in ruminants, which is the formation of stones in the urethra called calculi. The main mineral component of calculi from cattle and sheep is silica. “Pure” silica rock contains about 75% silica (that is, silicon dioxide) [17].
The purpose of this study is to determine the potential of rice bran and rice husks as feed and their impact on ruminants, as well as how to detect rice husk contamination in rice bran.
MATERIALS AND METHODS
This research is a literature study. Collection of publication data with the Publish or Perish version 8 application from various sources that match the theme. The citation process uses the Mendeley Desktop application version 1.19.8. [18].
RESULTS AND DISCUSSION
Rice seed structure
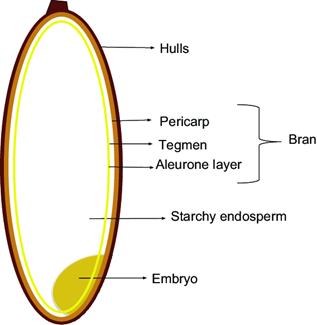
The grains of rice consist of endosperm, husks, bran, and contaminants, of which endosperm accounts for 70%, rice husks for 20% each, rice bran for 9% (Figure 1 and 2), and rice contamination for 1% of the total weight of the seed. During the production of milled rice, a large amount of rice husks is produced as a by-product [19]. Rice bran is rich in vitamins, minerals, essential fatty acids, dietary fibre, and other sterols [20]. Rice bran is not only a widely produced resource but also has high nutritional value. The nutrient content of rice bran is 12–16% protein, 12–23% fat, and 23–30% dietary fibre. In addition, it is rich in vitamins, minerals, and other nutritional factors such as phenantren, inositol, phytic acid, and glutamate [21].
Figure 1. Rice seed structure [22]
Figure 2. Constituent Components of Rice Seeds [23], [24]
Development of Ruminants in Indonesia
Livestock population is a collection or number of livestock that live in a certain area at a certain time. Based on the type, livestock are grouped into large livestock (beef cattle, dairy cattle, buffaloes, and horses), small livestock (goats, sheep, and pigs), poultry (free-range chickens, laying hens, broilers, ducks, and manila ducks), and various livestock (rabbits and quails). The largest populations of ruminants nationally in 2018–2022 are goats, beef cattle, sheep, and dairy cattle (Figure 3).
Figure 3. National Ruminant Livestock Population Based on Livestock Type in the Last 5 Years
Based on the figure above, there has been an increase in the number of beef cattle and dairy cattle in the last five years. This is due to the import of females and the success of the UPSUS SIWAB (Special Efforts for Compulsory Bunting Cows) program, where each parent cow is sought to be pregnant through a massive artificial insemination program by the government. The increase was also due to restrictions on community activities during the COVID-19 pandemic. Restrictions on religious activities such as Eid al-Fitr and Eid al-Adha for the Muslim community make slaughtering cows much less than normal. The price of beef cattle falling during the pandemic can cause huge losses if cattle are forced to be sold because most cattle farmers in Indonesia aim for investment. Slaughter of cattle for food industry production is greatly reduced because people greatly reduce the consumption of finished foods, such as meatballs, soto, gule, soup, rawon, satay, grilled meat, and others with basic ingredients of beef outside the home.
Meanwhile, with restrictions on community activities during the pandemic, the economic sector has decreased significantly, as a result of which people’s income has decreased. However, Muslims who are able to perform the qurban continue to perform their prayers, but because income decreases, many divert sacrificial animals from cows to sheep, so the sheep population decreases. It seems that the ruminant population shows a growing trend along with the needs of ruminants due to population growth.
The large ruminant population and the trend to increase from year to year will require a large amount of feed. Ruminant fodder, which is anything that can be eaten by livestock and does not pose any danger when livestock wear it, Examples of ruminant animal feed are grain, forage, and agricultural or livestock waste. The functions of feed include growth, basic life, production, and reproduction. [25]. In ruminant cultivation, feed contributes 63.52% of total production costs in forage beef cattle farms [26], 74% of total production is on dairy farms [27].
The distribution of ruminants, especially dairy cattle, beef cattle, goats, and sheep, in Indonesia, spread from Aceh to Papua, is presented in Figure 4. The highest population is found on the island of Java, namely in the provinces of West Java, East Java, and Central Java. This is because the island of Java has a large population, so the level of demand for meat is high. To meet these meat needs, almost all provinces in Java have become meat production centre in Indonesia.
Figure 4. National Ruminant Population (Dairy Cattle, Beef Cattle, Goats, and Sheep) by Province
The Importance of Feed to Ruminant Performance
Feed is a source of nutrients for animals. Feed includes pasture pastures, maintained forage crops, grain, plant residues, and by-products [28]. Feed is also everything that can be eaten by livestock, can be digested in whole or in part and does not interfere with the health of livestock [29].
The condition of livestock lacking balanced nutrients in feed is called malnutrition or undernutrition. Insufficient nutrient intake leads to poor reproductive performance, a slow growth rate, loss of body condition, increased susceptibility to parasites and diseases, and increased mortality [30], [31], [32] and increased enteric methane emissions [33].
Livestock responses to low nutrient quality will lead to loss of live weight, changes in metabolic and body composition, and death [34]. In periods of malnutrition, gluconeogenesis from propionate decreases due to a decrease in the availability of propionate. This condition is partially compensated by gluconeogenesis from amino acid proteolysis (AA), glycerol lipolysis, and lactic acid recycling. These metabolic changes are controlled by teleophoresis hormones such as insulin, glucagon, and norepinephrine, which, together with a decrease in splanchnic tissue mass and variations in body composition, result in a reduction in energy expenditure. Medium-term experiments (several weeks) show that dried offal, liver, and skeletal muscles contribute to changes in energy expenditure by 17–61%, 14–44%, and 5-7%, respectively [34]. When animals eat less or lack nutrients, they mobilize their body’s nutrient reserves, which are restored when given enough nutrients to sustain their reproductive cycle [35].
The term energy-protein malnutrition (PEM) has been used to describe inadequate feed provision, in terms of quantity, quality, or both, for ruminants [36]. The adaptation of ruminants to nutrient deficiency involves digestive, metabolic, and endocrine changes, which largely depend on the severity and duration of feed restriction [34]. Nutrient deficiencies affect reproductive function, interact with the seasons (photoperiods), and affect the state of the body [37]. When pregnancy occurs, if the supply of nutrients is not sufficient to meet the needs of the mother and fetus, the mother will begin to use her own reserves, leading to a decrease in the body condition score (BCS), which is an important determinant for optimal pregnancy [38].
The International Feed Identification System classifies feed ingredients based on the main nutrients provided by them. The system classifies feed ingredients into eight classes, namely: 1. dry forage and hay; 2. pasture, pasture plants, and fresh forage; 3. silage; 4. energy sources; 5. protein sources; 6. mineral sources; 7. sources of vitamins; and 8. additives [39].
Broadly speaking, feed for ruminants can be grouped into two categories: concentrate feed and fibrous feed [40]. Concentrate is a feed that serves to increase the nutritional value of a feed so that it can meet the needs of livestock, both for basic life, growth, production, and reproduction. Fibrous feed is the main feed given to livestock. Fibrous feed can be given as a single feed if the quality is high. Fibrous feed is usually added to concentrate feed because fibrous feed usually has a low nutrient quality [25].
Concentrates are foods that are low in crude fibre, contain a lot of BETN, and are very easy to digest [41]. Concentrates generally contain dry matter and food substances such as proteins, carbohydrates, fats, minerals, and vitamins. The application of concentrates depends on the quality of the forage given. The higher the quality of forage, the less food substances are supplied from concentrates; therefore, increasing livestock productivity may only be possible by giving high-quality concentrates [42]. Deterioration in feed quality can be caused by a long process experienced by feed from the factory to the farmer, temperature, humidity, and storage conditions [43].
The concentrate given to livestock can be a source of protein, a source of energy, or a combination of the two. Each concentrate has different characteristics, both in terms of nutrients, origin, economic value, and others. Protein sources for ruminants generally have a high nutritional value of protein, have higher degradability than forage groups, come more from food processing waste, and have a high price [44].
High-energy feed is concentrate feed containing a maximum of 20% crude fibre and 20% crude protein and a minimum of 2646 kcal of digestible energy (ED) per kg of air-dry feed (or a minimum of 60% of total digestible nutrients), plus one or more nutrients in high quantities that will increase nutritional adequacy [45]. One of the concentrate ingredients classified as energy source feed is rice bran, which is commonly found as animal feed. This feed ingredient is widely spread throughout Indonesia because it is related to the profession of most Indonesians, who are rice farmers.
The Potential of Rice Bran as Feed
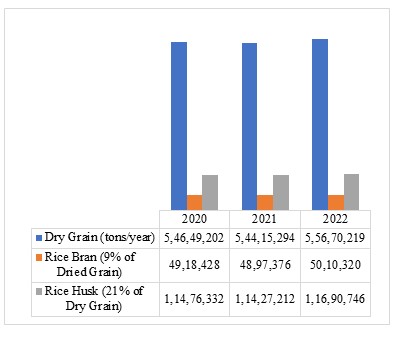
Rice bran is a concentrated energy source that can provide energy and protein from grass or forage given to ruminants. Rice bran is a by-product of the rice milling industry and constitutes about 9% of the total weight of coarse rice. Nationally, dry grain production experienced in 2022 was 55.67 million tons per year. This value will produce 5.01 million tons per year of rice bran and 11.69 million tons per year of rice husks. The value of national rice bran is very far from what is needed as a feed ingredient for ruminants, which reaches 19.88 million tons per year. From this data, it can be understood if it turns out that there is adulteration of rice bran mixed with rice husks (Figure 5).
Figure 5. Production of Dry Grain, Rice Bran and National Rice Husks in 2020-2022 (Tons)
The quality of rice bran as an animal feed ingredient is based on nutritional content and the presence or absence of other unwanted substances or ingredients, which are classified into three levels of quality, namely: quality I, quality II, and quality III. The quality requirements of rice bran as an animal feed ingredient are presented in Table 1 [46]:
Table 1. Quality of Rice Bran Based on Nutrient Content
| No. | Nutrient Content | Quality I | Quality II | Quality III |
| 1. | Water Content max (%) | 13 | 13 | 13 |
| 2. | Ash max (%) | 11 | 13 | 15 |
| 3. | Crude protein min (%) | 12 | 10 | 8 |
In terms of nutrients, rice bran is one of the main feed ingredients used in rice-producing countries and has the highest P-phytate content among plant-derived feed ingredients [47]. Rice bran contains P at 1.89%, but the availability of P in rice bran is greatly reduced due to its high phytate level. Phytate is a typical plant product formed during the seed ripening of plants as a form of primary phosphorus storage in cereal grains [48], [49]. About 80% of phytate is in the aleurone layer of cereal grains [50]. Phytate can be extracted from rice bran containing aleuron, pericarp, germ, and endosperm layers [51], [52]. Rice bran containing phytate can be used to produce phosphate starch [53].
Phosphorus is mainly stored in phytate form in plant seeds, potentially reducing its solubility in the rumen and its availability in ruminants, especially when the rumen is functioning less than optimally. The phytase enzyme catalyzes the gradual hydrolysis of phytate. In ruminants, there are five sources of enzymes available in the form of: rumen microbial phytase, endogenous mucosal phytase, colon microbial phytase, plant phytase, and exogenous microbial phytase [54].
The performance of young bulls with various levels of rice bran resulted in relatively similar weight gain for rice bran 20 and 30%. When the rate of rice bran is increased from 30% to 40% or 50%, weight gain is significantly decreased, and feed utilization is less efficient. The reduced weight gain and feed efficiency may be offset by the low cost of rice bran. However, if the administration is high, it will cause diarrhea when the use of rice bran in the ration is 40–50% [55]. The use of 9% rice bran in dairy cows can be replaced entirely with cassava skin as an energy source [56]. The use of 20% rice bran in concentrate can be replaced with tamarind seed flour in dairy cows [57]. The increasing use of rice bran substituted with oil palm meal by 25–15% can increase the final weight of thin-tailed sheep [58]. The use of 33.98%–38.84% rice bran in substituted concentrate with 14.5–7.71% beer raw material grains does not adversely affect peanut and goat methane emissions [59]. The use of up to 15% rice bran tends to increase feed consumption and goat body weight [60]. In Table 2, the results of research on the use of rice bran as ruminant feed are presented.
Table 2. The use of rice bran in ruminants
| No. | Reference | Use of Rice Bran in Concentrate (%) | Livestock |
| 1 | [61] | 24 | Garut Sheep |
| 2 | [62] | 14,41 | Garut Sheep |
| 3 | [63] | 12 | Garut Sheep |
| 4 | [64] | 70 | Beef |
| 5 | [65] | 40 | Bali Catlle |
| 6 | [66] | 18 | Goat |
| 7 | [67] | 37 | Goat |
| 8 | [68] | 25 | Dairy Goat |
| 9 | [69] | 9,7 | Dairy |
| 10 | [70] | 9 | Dairy |
Based on the data above (Table 2), the use of rice bran concentrate as feed for sheep ranges from 12–41%, for dairy goats, 18–37%, for beef cattle, 40–70%, and for dairy cattle, 9–9.7%. This data shows that rice bran is commonly used in ruminant rations, especially in Indonesia. The need for rice bran is very high, but supplies are less than needed. Therefore, rice bran is often supplemented with various other ingredients, and the most commonly used is rice husks.
Impact of Rice Husks in Rations on Livestock Performance
Rice husks are an important byproduct of the rice milling process, with very large quantities produced worldwide each year. During the production of milled rice, a large amount of 20–21% rice husks is produced as a by-product [19].
Rice husks contain 24% cellulose, 12% hemicellulose, and 4.5% lignin components, so they can be recommended to be given to ruminants who are more able to utilize insoluble carbohydrate components. In addition, rice husks contain 14.4–23.4% glucose and 12-28.6% silos, which are the largest components in rice husks. Rice husks are agricultural by-products that can still be used as feed and are even categorized as an alternative energy source for livestock [71]. Although it has compounds that can be used as nutrients for ruminants, rice husks, in addition to containing lignin, also contain silica [19].
Rice husks are lignocellulose biomass waste rice milling products. The use of rice husks in rations is very limited due to the presence of lignin compounds. The molecular structure of lignin complexes in lignocellulose material will prevent degradation by rumen microbial enzymes [72]. These compounds are difficult to degrade by rumen microbes, so rice husks are difficult to digest by the digestive tract.
When the lignin content is low, the rate of degradation increases, but when the lignin content increases, the rate of degradation decreases. This suggests that lignin content is the limiting factor in determining the degree of degradation of the lignin-hemicellulose complex [73]. The high content of lignin in corn cobs also results in a slow rate of feed release, resulting in a low rate of rumen microbial degradation and a decrease in microbial populations and N supply in ruminants.
If the content of foodstuffs is high, especially lignin, then the digestibility coefficient of foodstuffs is low. High crude fibre content in a ration generally does not support livestock production due to low ration consumption. This is because the high crude fibre content in the feed inhibits the movement of digesta in the digestive tract [74]. Most cellulose and hemicellulose are easily digested by rumen microbes, but lignin is in lignocellulose and lignohemicellulose complex bonds, making it difficult to digest [74].
The effect of lignin on the rumen is the occurrence of a higher incidence of lesions on the rumen wall originating from animals receiving lignin. In the pure lignin treatment, four animals (50%) had lesions with a score of 3 or higher, and two of these animals (25% of the total) achieved a score of 5 (injury to 50% of the rumen). Six animals fed pure lignin had short-term diarrhea, suggesting possible acidosis [75]. The tolerance of cattle to lignin in the ration is a maximum of 7% [76].
In addition to lignin, rice husks contain high silica and have an impact on ruminants. The presence of silica in meadow plants ensures that grazing ruminants ingest large enough amounts of silica, most of which is in solid form. Apart from being slightly soluble, this silica does not change as it passes through the gastrointestinal tract, and its known effects on animals are physical or mechanical. Dissolved silica absorbed from the gastrointestinal tract is carried to the kidneys and excreted in the urine. Although usually easily excreted, silica is sometimes stored to form rocks or uroliths [77].
The impact of silica on ruminants can cause wear and tear on teeth because silica is harder than dental tissue [78], affects the histological structure of the rumen wall due to physical abrasion. In old sheep, the walls of the rumen sometimes have extensive fine patches, in which papillae greatly decrease in size [79], decrease digestibility since silica, like lignin, is an integral part of the plant cell wall matrix and can also reduce the accessibility of cell wall carbohydrates to attack by digestive microorganisms [77], and lowers the palatability of feed as the leaf surface becomes rougher to the touch [80], So that grass will be less pleasant to eat for grazing animals. The reduction in silica content to less than 10% dry weight increased the digestibility of dry matter in vivo straw in both varieties and improved the digestibility of organic matter in vitro rice straw IR36 [81].
Rice Bran Adulteration Testing Technique
The high need for rice bran for animal feed, which is inversely proportional to production, causes prices to be expensive. This causes the adulteration of rice bran to occur quite frequently by mixing rice bran with other materials that have physical characteristics almost the same as the original raw materials to be faked. One of the materials used as a counterfeiting material for rice bran is milled husks [82], peanut shell flour [83], corn cob flour [84] and sawn powder [85].
Bran testing is carried out using physical (organoleptic) test methods and chemical analysis. Chemical analysis shows high accuracy of results but takes a long time and is expensive, while organoleptic tests take place quickly (rapid tests) and are cheaper but less accurate. The analysis of rice husk content in rice bran was carried out using the phloroglucinol-HCl test. Rice husks with high lignin content will have a red color response [46]. The bran adulteration test to date, carried out by physical and chemical test methods, is presented in the following Table 3.
Table 3. Test Method of Various Contaminants in Rice Bran
| No. | Reference | Material | Contaminants | Method |
| 1 | [86] | Rice Bran | Rice Husk, Limestone, Sand, Cassava Waste | Rapid Test Husk (lignin) using Phloroglucinol, Limestone using “Carbonate Test”, Sand and Zeolite using “floatation method”, cassava waste using iodine “spot test” |
| 2 | [87] | Rice Bran | Rice Husk | Rice bran is analyzed for its chemical composition (proximate analysis) and physical characteristics (density test, density compaction test, total solubility test and phloroglucinol test) |
| 3 | [84] | Rice Bran | Corn cob flour | Using physical test of specific density, bulk density, solid bulk solid angle repose, particle size increase |
| 4 | [85] | Rice Bran | Sawdust Powder | The value of physical properties of rice bran is specific gravity, density, solid bulk solid, pile angle |
| 5 | [88] | Rice Bran | – | The physical property modifiers observed in this experiment were stack density, stack compaction density, specific gravity and moisture content. The chemical property modifiers observed were crude protein content, gross energy and crude fibre. |
| 6 | [89] | Rice Bran | – | Assessing the nutrient content of rice bran through physical tests and compared with chemical test methods |
| 7 | [90] | Rice Bran | Rice Husk | Ultrasonic wave frequency 185 kHz, attenuation parameters and velocity |
| 8 | [91] | Rice Bran | Rice Husk | Determine the quality of good rice bran through the physical characteristics of rice bran by conducting organoleptic tests (colour, aroma, texture, insects), husk tests and pile density tests. |
| 9 | [92] | Rice Bran | Rice Husk | Measuring the accuracy of training, validation, and test data on rice bran adulteration with husks using CNN-based Image Analysis and using phloroglucinol |
| 10 | [82] | Rice Bran | Rice Husk | Physical and chemical properties, observed parameters are stack density, stack compaction density, stack angle, moisture content, ash, crude protein, crude fat, crude fibre and phloroglucinol test. |
CONCLUSIONS
Rice bran is a very potential feed ingredient for ruminants because it contains nutrients that are in accordance with their needs. The high need for rice bran is not in line with its productivity, so it is mixed with many other materials, especially rice husks. Mixing with other materials can be determined by their physical and chemical properties. Rice husks are an alternative energy source for ruminants. To improve the digestibility of rice husks through physical, chemical, and biological treatment. Recommendations for using rice husks as a feed ingredient have not been widely reported, so future research is needed.
ACKNOWLEDGMENT
This research is supported by a grant from the Padjadjaran University Doctoral Research Scheme for Padjadjaran University Lecturers (RDDU) in 2023.
REFERENCES
- BPS, “Jawa Barat dalam Angka 2022,” Badan Pusat Statistik Provinsi Jawa Barat, 2022.
- Bakri, “Komponen Kimia dan Fisika Abu Sekam Padi Sebagai SCM untuk Pembuatan Komposit Semen,” Perennial, Volume 5 No. 1, 2008.
- R. Rinaldi et al., “Paving the way for lignin valorisation: recent advances in bioengineering, biorefining and catalysis,” Angewandte Chemie International Edition, vol. 55, no. 29, pp. 8164–8215, 2016.
- J. Zakzeski, P. C. A. Bruijnincx, A. L. Jongerius, and B. M. Weckhuysen, “The catalytic valorization of lignin for the production of renewable chemicals,” Chem Rev, vol. 110, no. 6, pp. 3552–3599, 2010.
- C. B. Tucker, D. M. Weary, and D. Fraser, “Effects of three types of free-stall surfaces on preferences and stall usage by dairy cows,” J Dairy Sci, vol. 86, no. 2, pp. 521–529, 2003.
- R. L. Belyea, M. B. Foster, and G. M. Zinn, “Effect of delignification on in vitro digestion of alfalfa cellulose,” J Dairy Sci, vol. 66, no. 6, pp. 1277–1281, 1983.
- A. M. Atuhaire, F. Kabi, S. Okello, S. Mugerwa, and C. Ebong, “Optimizing bio-physical conditions and pre-treatment options for breaking lignin barrier of maize stover feed using white rot fungi,” Animal Nutrition, vol. 2, no. 4, pp. 361–369, 2016.
- W. Widodo, F. Wahyono, and S. Sutrisno, “Kecernaan bahan kering, kecernaan bahan organik, produksi VFA dan NH3 pakan komplit dengan level jerami padi berbeda secara in vitro,” Animal Agriculture Journal, vol. 1, no. 1, pp. 215–230, 2012.
- A. B. Prasetyo, B. I. M. Tampoebolon, and L. K. Nuswantara, “Kandungan Serat Kasar, Kecernaan Serat Kasar, dan Fermentabilitas Bonggol Singkong yang Difermentasi Menggunakan Aspergillus niger,” Jurnal Agripet, vol. 22, no. 2, pp. 204–212, 2022.
- E. Wijayanti, F. Wahyono, and S. Surono, “Kecernaan nutrien dan fermentabilitas pakan komplit dengan level ampas tebu yang berbeda secara in vitro,” Animal Agriculture Journal, vol. 1, no. 1, pp. 167–179, 2012.
- S. A. Akbar, “Pemanfaatan tandan kosong sawit fermentasi yang dikombinasikan dengan defaunasi dan protein by pass rumen terhadap performans ternak domba,” Jurnal Pengembangan Peternakan Tropis, vol. 32, no. 2, pp. 80–85, 2007.
- S. Subaer, “Pengantar Fisika Geopolimer,” 2015.
- J. Lee et al., “Biokinetics of food additive silica nanoparticles and their interactions with food components,” Colloids Surf B Biointerfaces, vol. 150, pp. 384–392, 2017.
- D. J. McClements, H. Xiao, and P. Demokritou, “Physicochemical and colloidal aspects of food matrix effects on gastrointestinal fate of ingested inorganic nanoparticles,” Adv Colloid Interface Sci, vol. 246, pp. 165–180, 2017.
- A. Agbagla-Dohnani, P. Noziere, B. GAILLARD-MARTINIE, M. Puard, and M. Doreau, “Effect of silica content on rice straw ruminal degradation,” J Agric Sci, vol. 140, no. 2, pp. 183–192, 2003.
- P. J. Van Soest, Nutritional ecology of the ruminant. Cornell university press, 2018.
- C. B. Bailey, “SILICA METABOLISM AND SILICA UROLITHIASIS IN RUMINANTS: A REVIEW,” Can J Anim Sci, vol. 61, no. 2, pp. 219–235, Jun. 1981, doi: 10.4141/cjas81-031.
- U. Rosani, “Analisis Bibliometrik Perkembangan Penelitian Feed Additive Pada Ternak Ruminansia Dengan Vosviewer,” Jurnal Nutrisi Ternak Tropis dan Ilmu Pakan, vol. 3, no. 3, 2022, doi: 10.24198/jnttip.v3i3.35992.
- Y. Zou and T. Yang, “Chapter 9 – Rice Husk, Rice Husk Ash and Their Applications,” L.-Z. Cheong and X. B. T.-R. B. and R. B. O. Xu, Eds., AOCS Press, 2019, pp. 207–246. doi: https://doi.org/10.1016/B978-0-12-812828-2.00009-3.
- K. Gul, B. Yousuf, A. K. Singh, P. Singh, and A. A. Wani, “Rice bran: Nutritional values and its emerging potential for development of functional food – A review,” Bioactive Carbohydrates and Dietary Fibre, vol. 6, no. 1. pp. 24–30, 2015. doi: 10.1016/j.bcdf.2015.06.002.
- M. Sohail, A. Rakha, M. S. Butt, M. J. Iqbal, and S. Rashid, “Rice bran nutraceutics: A comprehensive review,” Crit Rev Food Sci Nutr, vol. 57, no. 17, pp. 3771–3780, 2017, doi: 10.1080/10408398.2016.1164120.
- S. Cozzano, A. Fernández, M. Castillo, and A. Fernández, “New Functional Ingredients From Agroindustrial By-Products for the Development of Healthy Foods,” 2018. doi: 10.1016/B978-0-08-100596-5.22140-9.
- A. R. Bodie, A. C. Micciche, G. G. Atungulu, M. J. Rothrock Jr, and S. C. Ricke, “Current trends of rice milling byproducts for agricultural applications and alternative food production systems,” Front Sustain Food Syst, vol. 3, p. 47, 2019.
- P. Limtrakul, W. Semmarath, and S. Mapoung, “Anthocyanins and Proanthocyanidins in Natural Pigmented Rice and Their Bioactivities,” in Phytochemicals in Human Health, V. Rao, D. Mans, and L. Rao, Eds., Rijeka: IntechOpen, 2020, p. Ch. 1. doi: 10.5772/intechopen.86962.
- Y. Daskunda, G. Joseph, and I. Sangaji, “Analisis Kandungan Nutrisi Lamtoro Mineral Blok (LMB) Sebagai Pakan Tambahan pada Ternak Ruminansia,” JURNAL PERTANIAN KEPULAUAN, vol. 4, no. 2, pp. 52–60, 2020.
- K. L. Nuhon and L. F. Hetharia, “Analisis Tingkat Pendapatan Usaha Ternak Sapi Potong Sistem Gaduhan Di Distrik Arso Kabupaten Keerom Provinsi Papua,” Jurnal Pertanian Terpadu Santo Thomas Aquinas, vol. 1, no. 2, pp. 35–40, 2022.
- D. A. Candra and R. Anggriawan, “ANALYSIS BIAYA PRODUKSI USAHA TERNAK SAPI PERAH ‘ANUGERAH’ DI KECAMATAN PAGU KABUPATEN KEDIRI,” Jurnal Agriovet, vol. 3, no. 1, pp. 1–10, 2020.
- M. F. Fuller, “The encyclopedia of farm animal nutrition: AE.,” in The encyclopedia of farm animal nutrition, CABI publishing Wallingford UK, 2004, pp. 1–198.
- D. A. Lubis, “Ilmu makanan ternak umum,” Penerbit PT. Pembangunan. Jakarta, 1992.
- E. Charmley, M. L. Stephens, and P. M. Kennedy, “Predicting livestock productivity and methane emissions in northern Australia: development of a bio-economic modelling approach,” Aust J Exp Agric, vol. 48, no. 2, pp. 109–113, 2008.
- R. L. McCown, “The climatic potential for beef cattle production in tropical Australia: Part I—Simulating the annual cycle of liveweight change,” Agric Syst, vol. 6, no. 4, pp. 303–317, 1981.
- J. Kanani, S. D. Lukefahr, and R. L. Stanko, “Evaluation of tropical forage legumes (Medicago sativa, Dolichos lablab, Leucaena leucocephala and Desmanthus bicornutus) for growing goats,” Small Ruminant Research, vol. 65, no. 1–2, pp. 1–7, 2006.
- K. A. Johnson and D. E. Johnson, “Methane emissions from cattle,” J Anim Sci, vol. 73, no. 8, pp. 2483–2492, 1995.
- Y. Chilliard, F. Bocquier, and M. Doreau, “Digestive and metabolic adaptations of ruminants to undernutrition, and consequences on reproduction,” Reprod Nutr Dev, vol. 38, no. 2, pp. 131–152, 1998.
- Y. Chilliard, A. Ferlay, Y. Faulconnier, M. Bonnet, J. Rouel, and F. Bocquier, “Adipose tissue metabolism and its role in adaptations to undernutrition in ruminants,” Proceedings of the Nutrition Society, vol. 59, no. 1, pp. 127–134, 2000, doi: DOI: 10.1017/S002966510000015X.
- G. R. Oetzel, “Protein-energy malnutrition in ruminants.,” Vet Clin North Am Food Anim Pract, vol. 4, no. 2, pp. 317–329, 1988.
- Y. Chilliard, F. Bocquier, and M. Doreau, “Undernutrition adaptation of ruminants,” Cahiers de Nutrition et de Dietetique (France), 1998.
- L. Clarke, L. Heasman, D. T. Juniper, and M. E. Symonds, “Maternal nutrition in early-mid gestation and placental size in sheep,” British Journal of Nutrition, vol. 79, no. 4, pp. 359–364, 1998.
- L. C. Kearl, International Feed Databank System: Coding Instructions and Processing Procedures. in International Network of Feed Information Centers publication. INFIC, 1980. [Online]. Available: https://books.google.co.id/books?id=Fw5JAAAAYAAJ
- J. Blakely and D. H. Bade, “Ilmu Peternakan (Animal Sicence).” Gadjah Mada University Press, Yogyakarta, 1998.
- H. S. Hartadi, A. D. Tillman, S. Reksohadiprojo, S. Prawirokusumo, and S. Lebdosoekojo, “Ilmu Makanan Ternak Dasar Cetakan Ke-6.” Gadjah Mada University Press, Yogyakarta, 1998.
- S. B. Siregar, “Ransum ternak ruminansia,” Penebar Swadaya. Jakarta, vol. 16, 1994.
- P. Dwi Kusuma, S. Syamsuhaidi, and M. Kurniawan, “Evaluasi Kualitas Pakan Komplit dan Konsentrat Unggas yang Diperdagangkan di Kota Mataram,” Jurnal Peternakan Sriwijaya, vol. 5, no. 1, pp. 30–38, 2016.
- A. N. Syamsi, H. S. Widodo, Y. Subagyo, and P. Soediarto, “INDEKS SINKRONISASI PROTEIN-ENERGI DARI BEBERAPA KONSENTRAT SUMBER PROTEIN BAGI RUMINANSIA,” in PROSIDING SEMINAR NASIONAL TEKNOLOGI AGRIBISNIS PETERNAKAN (STAP), 2021, pp. 244–251.
- W. G. Pond, D. E. Ullrey, and C. K. Baer, Encyclopedia of Animal Science-(Two-Volume Set). CRC Press, 2018.
- SNI, “Dedak Padi-Bahan pakan Ternak 3178,” Jakarta (ID): Badan Standarisasi Nasional, 2013.
- A. Mohebbifar, M. Torki, and H. A. Ghasemi, “Effect of phytase supplementation of diets with different levels of rice bran and non-phytate phosphorus on productive performance, egg quality traits, leukocytes profile and serum lipids of laying hens reared indoor under high environmental temperatures,” Anim Feed Sci Technol, vol. 207, pp. 222–233, 2015, doi: https://doi.org/10.1016/j.anifeedsci.2015.06.024.
- F. Loewus, “Biosynthesis of phytate in food grains and seeds,” Food phytates, pp. 53–61, 2002.
- G. T. Tsao, Y. Zheng, J. Lu, and C. S. Gong, “Adsorption of heavy metal ions by immobilized phytic acid,” Appl Biochem Biotechnol, vol. 63, pp. 731–741, 1997.
- B. L. O’Dell, A. R. De Boland, and S. R. Koirtyohann, “Distribution of phytate and nutritionally important elements among the morphological components of cereal grains,” J Agric Food Chem, vol. 20, no. 3, pp. 718–723, 1972.
- R. M. Saunders, “Rice bran: Composition and potential food uses,” Food Reviews International, vol. 1, no. 3, pp. 465–495, 1985.
- N. V Makarenko, L. A. Zemnukhova, A. V Nemtarev, A. V Kovekhova, and O. D. Arefieva, “Composition and structure of phytic acid derivatives from rice bran,” Bioresources, vol. 13, no. 2, pp. 3411–3419, 2018.
- Lee, S.-R. Kim, J. Y. Park, and E. Y. Park, “Phytate-mediated phosphorylation of starch by dry heating with rice bran extract,” Carbohydr Polym, vol. 282, p. 119104, 2022, doi: https://doi.org/10.1016/j.carbpol.2022.119104.
- E. Humer, “Phytate in feed ingredients and potentials for improving the utilization of phosphorus in ruminant nutrition,” Animal Feed Science and Technology, vol. 209. pp. 1–15, 2015. doi: 10.1016/j.anifeedsci.2015.07.028.
- T. W. White, “Rice by-products in ruminant rations,” 1985.
- F. Agustin, Erpomen, and R. W. S. Ningrat, “The Use of Cassava Peel as a Source of Energy for Substituting Rice Bran in Ration Containing Gliricidia maculata Leaves in Dairy Cows,” in IOP Conference Series: Earth and Environmental Science, IOP Publishing, 2020, p. 12077. doi: 10.1088/1755-1315/478/1/012077.
- A. Nunoi, M. Wanapat, S. Foiklang, T. Ampapon, and B. Viennasay, “Effects of replacing rice bran with tamarind seed meal in concentrate mixture diets on the changes in ruminal ecology and feed utilization of dairy steers,” Trop Anim Health Prod, vol. 51, no. 3, pp. 523–528, 2019, doi: 10.1007/s11250-018-1719-z.
- T. Suteky, I. Lestari, M. Sari, and M. Manurung, “The Digestibility of Different Level of Palm Kernel Cake and Rice Bran Supplementation in Sheep,” in International Seminar on Promoting Local Resources for Sustainable Agriculture and Development (ISPLRSAD 2020), Atlantis Press, 2021, pp. 172–175.
- Y. Yanti, W. A. Pamungkas, F. Hermanu, S. Putranto, S. D. Widyawati, and W. P. S. Suprayogi, “Effects of different composition of brewer grain and rice bran in the concentrate on methane emission of Kacang goat,” in IOP Conference Series: Earth and Environmental Science, IOP Publishing, 2021, p. 12078.
- N. H. Davalos and R. A. Quitain, “Coconut Meat and Rice Bran as Feed for Goat (Capra Hircus),” Sciences, vol. 13, no. 1, pp. 647–650, 2022.
- S. A. Yamashita, R. D. Rachmat, A. R. Tarmidi, B. Ayuningsih, and I. Hernaman, “Kecernaan ransum yang mengandung limbah roti pada domba,” J. Ilmu dan Teknologi Peternakan Tropis, vol. 7, no. 1, pp. 47–51, 2020.
- I. Hernaman et al., “Perbandingan model pendugaan total digestible nutrients (tdn) dan protein tercerna pada domba garut jantan yang diberi ransum berbasis bahan pakan lokal,” Jurnal Agripet, vol. 19, no. 1, pp. 1–6, 2019.
- B. Ayuningsih, A. Rochana, I. Hernaman, H. Hidayat, and T. Dhalika, “Kadar NPK feses domba garut yang diberi ransum mengandung silase daun rami (Boehmeria nivea),” Jurnal Ilmu dan Teknologi Peternakan Tropis, vol. 6, pp. 161–165, 2019.
- A. Supriyantono, D. A. Iyai, and A. R. Ollong, “Peningkatan Produktivitas Sapi Potong Melalui Introduksi Pakan Konsentrat Dengan Bahan Lokal Pada Masyarakat Asli Papua: Productivity Improvement of Beef Cattle through the Introduction of Feed Concentrates to the Local Papuan,” IGKOJEI: Jurnal Pengabdian Masyarakat, vol. 1, no. 1, pp. 21-â, 2020.
- M. R. Soli, Y. U. L. Sobang, and M. Yunus, “Pengaruh Suplementasi Konsentrat Limbah Isi Rumen Sapi Fermentasi Level Berbeda Terhadap Kinerja Produksi Sapi Bali Penggemukan: The Impact of Fermented Waste Contents in Concentrated Feed Supplementation Cattle Rumen at Different Levels on Production Per,” Jurnal Peternakan Lahan Kering, vol. 4, no. 4, pp. 2387–2393, 2022.
- M. Kristofer, M. S. Abdullah, and J. N. Kihe, “Subtitusi Pakan Konsentrat Mengandung Tepung Daun Binahong (Anredera cordifolia ten, Steeni) Dalam Ransum Terhadap Status Fisiologis Kambing Lokal Jantan: Substition Concentrated Feed Containing Binahong Leaves (Anredera cordifoliaten, Steeni) in Rationon,” Jurnal Peternakan Lahan Kering, vol. 3, no. 3, pp. 1602–1607, 2021.
- E. Wea and D. Amalo, “Pengaruh substitusi dedak padidengan kulit buah kopi terfermentasi aspergillus niger dalam konsentrat terhadap performans kambing,” Jurnal Peternakan Lahan Kering, vol. 1, no. 2, pp. 212–221, 2019, [Online]. Available: http://publikasi.undana.ac.id/index.php/JPLK/article/view/k279/278
- E. Sulistyowati, I. Badarina, S. Mujiharjo, R. Mariska, and E. Khoirul, “Analisis Ekonomis Penggunaan Tepung Kulit Durian Fermentasi dalam Konsentrat untuk Kambing Perah,” Jurnal Sain Peternakan Indonesia, vol. 15, no. 1, pp. 37–42, 2020.
- H. A. Hudori, H. Rujito, M. Muksin, F. E. Ardhi Pratama, and P. Andini, “Formulasi Ransum Alternatif Untuk Meningkatkan Efisiensi Usaha Peternakan Sapi Perah (Studi Kasus pada Peternakan Bestcow Farm Jember),” Jurnal Ilmu Peternakan Terapan, vol. 3, no. 2, pp. 67–73, Jul. 2020, doi: 10.25047/jipt.v3i2.1956.
- E. Pangestu, “Peningkatan ketrampilan peternak dalam pembuatan pakan konsentrat sapi perah di KT Tunas Muda Desa Karangnongko Kecamatan Mojosongo Kabupaten Boyolali,” in Seminar Nasional Pengabdian Kepada Masyarakat UNDIP 2020, 2020.
- W. G. Piliang, “Strategi Penyediaan Pakan Ternak Berkelanjutan Melalui Pemanfaatan Energi Alternatif,” Orasi Ilmiah Guru Besar Tetap Ilmu Nutrisi, Fapet IPB, Bogor, 1997.
- M. Khatoon, S. J. Jakhesara, D. N. Rank, C. G. Joshi, and A. P. Kunjadiya, “Exploration of rumen microbial and carbohydrate-active enzyme profiles in cattle fed coir a lignin-rich diet using a metagenomic approach,” Gene, vol. 846, p. 146868, 2022, doi: https://doi.org/10.1016/j.gene.2022.146868.
- R. E. Brice and I. M. Morrison, “The degradation of isolated hemicelluloses and lignin-hemicellulose complexes by cell-free, rumen hemicellulases,” Carbohydr Res, vol. 101, no. 1, pp. 93–100, 1982, doi: https://doi.org/10.1016/S0008-6215(00)80797-1.
- T. Lestari and L. K. Nuswantara, “Degradasi Bahan Kering dan Bahan Organik dengan Berbagai Level Jerami Padi secara In Sacco pada Kambing Jawarandu,” Animal Agriculture Journal, vol. 1, no. 1, pp. 875–888, 2012.
- H. V. A. Bezerra, S. B. Gallo, A. F. Rosa, A. C. Fernandes, S. da L. e Silva, and P. R. Leme, “Impact of purified lignin on performance, rumen health, oxidative stress control and meat quality of lambs fed a high-concentrate diet,” Livest Sci, vol. 231, p. 103882, 2020.
- A. L. Darmawan et al., “The study on in vitro digestibility of soaked palm oil fiber by filtrated palm oil fruit bunch ash,” Majalah Ilmiah Peternakan, vol. 17, no. 1, p. 164265, 2014.
- L. H. P. Jones and K. A. Handreck, “Silica In Soils, Plants, and Animals,” vol. 19, A. G. B. T.-A. in A. Norman, Ed., Academic Press, 1967, pp. 107–149. doi: https://doi.org/10.1016/S0065-2113(08)60734-8.
- G. Baker, L. H. P. Jones, and I. D. Wardrop, “Cause of wear in sheeps’ teeth,” Nature, vol. 184, no. 4698, pp. 1583–1584, 1959.
- G. Baker and L. Jones, “LHP & Wardrop, ID 1961,” Aust. J. agric. Res, vol. 12, p. 462, 1961.
- L. H. P. Jones and K. A. Handreck, “Studies of silica in the oat plant: III. Uptake of silica from soils by the plant,” Plant Soil, vol. 23, pp. 79–96, 1965.
- M. L. F. C. Balasta, C. M. Perez, B. O. Juliano, C. P. Vlllareal, J. N. A. Lott, and D. B. Roxas, “Effects of silica level on some properties of Oryza sativa straw and hull,” Canadian Journal of Botany, vol. 67, no. 8, pp. 2356–2363, Aug. 1989, doi: 10.1139/b89-301.
- A. M. Rusyidi, Evaluasi Kualitas Dedak Padi Sebagai Bahan Pakan Yang Terkontaminasi Sekam Padi= Evaluation Of The Quality Of Rice Bran As a Feed Ingredients Contaminated …. repository.unhas.ac.id, 2022. [Online]. Available: http://repository.unhas.ac.id/id/eprint/13229/
- M. Ridla and A. Rosalina, Evaluasi Pemalsuan Dedak Padi Dengan Penambahan Tepung Kulit Kacang Tanah Menggunakan Uji Fisik. agris.fao.org, 2014. [Online]. Available: https://agris.fao.org/agris-search/search.do?recordID=ID2021102014
- N. S. Alhasanah, M. Ridla, and A. D. Lubis, “Evaluasi Pemalsuan Dedak Padi dengan Penambahan Tepung Tongkol Jagung Menggunakan Uji Fisik,” Repository.Ipb.Ac.Id, 2014, [Online]. Available: https://repository.ipb.ac.id/handle/123456789/68232
- Y. D. Istikhodriah, M. Ridla, and A. D. Lubis, “Evaluasi Pemalsuan Dedak Padi dengan Penambahan Serbuk Gergaji Menggunakan Uji Fisik,” Bogor (ID): Institut Pertanian Bogor. 2014. [Online]. Available: https://agris.fao.org/agris-search/search.do?recordID=ID2021102014
- W. Myint, “Check point determination for contamination and adulteration of rice bran.,” Proceedings of the Annual Research Conference …, 2005, [Online]. Available: https://www.cabdirect.org/cabdirect/abstract/20143398203
- A. H. Wibowo, Nahrowi, and I. G. Permana, Predicting rice bran nutrients based on physical characteristics. agris.fao.org, 2010. [Online]. Available: https://agris.fao.org/agris-search/search.do?recordID=ID2021109959
- C. Hidayat, Sumiati, and S. Iskandar, “Kualitas Fisik dan Kimiawi Dedak Padi yang Dijual di Toko Bahan Pakan di Sekitar Wilayah Bogor,” Prosiding Seminar Nasional Teknologi Peternakan dan …. 2015. [Online]. Available: http://medpub.litbang.pertanian.go.id/index.php/semnas-tpv/article/view/2447
- S. Ansor, M. Ridla, and A. Jayanegara, “Evaluasi Uji Fisik Kualitas Dedak Padi Di Kabupaten Kebumen Jawa Tengah,” Repository.Ipb.Ac.Id. pp. 1–32, 2015. [Online]. Available: https://repository.ipb.ac.id/handle/123456789/78084
- D. D. Bhagaspati, N. S. Syafei, and D. Hidayat, “Investigation of Husk-Adulterated Rice Bran Using Ultrasonic Wave,” in AIMS 2021 – International Conference on Artificial Intelligence and Mechatronics Systems, IEEE, Apr. 2021, pp. 1–5. doi: 10.1109/AIMS52415.2021.9466011.
- M. D. Nugroho, L. Liman, R. Sutrisna, and M. Muhtarudin, “Uji Kualitas Dedak Padi Di Kabupaten Lampung Tengah,” Jurnal Riset dan Inovasi Peternakan (Journal of Research and Innovation of Animals), vol. 6, no. 3, pp. 286–292, 2022, doi: 10.23960/jrip.2022.6.3.286-292.
- H. Rizqi, Albarki, A. Tata, Permana, and A. Jayanegara, Testing of Counterfeiting Rice Bran With Rice Husk Using Image Analysis Method. agris.fao.org, 2022. [Online]. Available: https://agris.fao.org/agris-search/search.do?recordID=ID2022100046


![Constituent Components of Rice Seeds [23], [24]](https://rsisinternational.org/journals/ijrsi/wp-content/uploads/2024/02/2-2.png)