Investigation of the Subchronic Toxicity of Graded Doses of Extracts of Dialium guineense Stem Bark in Rats
- Abu O.D.
- Awhin, E.P.
- Enadeghe, R.
- Eze-Nwaobasi, O.P.
- 15-23
- Jan 27, 2024
- Toxicology
Investigation of the Subchronic Toxicity of Graded Doses of Extracts of Dialium guineense Stem Bark in Rats
Abu O.D.1*, Awhin, E.P.2, Enadeghe, R.1 and Eze-Nwaobasi, O.P.1
1Department of Biochemistry, Faculty of Life Sciences, University of Benin, Benin City, Nigeria.
2Department of Medical Biochemistry, Faculty of Basic Medical Sciences, Delta State University, Abraka, Delta State, Nigeria.
*Corresponding Author
DOI: https://doi.org/10.51244/IJRSI.2024.1101002
Received: 09 December 2023; Revised: 19 December 2023; Accepted: 25 December 2023; Published: 26 January 2024
ABSTRACT
Aim: To investigate the subchronic toxicity of graded doses of aqueous and ethanol extracts of D. guineense stem bark in rats. Materials and Methods: A total of seventy (70) rats (35 per extract) weighing 160 to 180 g (mean weight = 170 ± 10 g) were divided into seven (7) groups of five (5) rats each. Group I served as control, while rats in groups II – VII received graded doses of extract (200 – 5000 mg/kg body weight, bwt) for a duration of 28 days. Weight parameters, liver malondialdehyde (MDA) level and histopathological assessment were carried out. Results: Percentage increases in body weights of rats treated with aqueous or ethanol extract of D. guineense stem bark were significantly reduced, when compared with control group (p < 0.05), but there were no significant differences in the corresponding relative organ weights and tissue MDA level, among the groups (p > 0.05). Similarly, the extracts did not significantly alter the normal architecture of the liver, but induced mild periportal lymphocytosis and kupffer cell activation. Conclusion: The aqueous and ethanol extracts of the medicinal plant stem bark did not elicit any deleterious effects on rat liver.
Keywords: Dialium guineense, Graded doses, Hepatotoxicity, Malondialdehyde, Tissue histology.
INTRODUCTION
As the largest important organ in the human body, the liver is the site for essential biochemical reactions. It functions in the detoxification of toxic substances and synthesis of useful biomolecules [1]. Therefore, damage to the liver leads to grave consequences. Liver damage may results from chronic alcohol abuse, viral hepatitis or inherited metabolic disease [2, 3]. Liver damage is characterized by cellular necrosis, fibrosis, increase in tissue lipid peroxidation and depletion in tissue glutathione level [4]. Most hepatotoxic chemicals damage liver cells via induction of lipid peroxidation and other oxidative processes [5].
Herbal medicines derived from plant sources are being increasingly utilized to treat a variety of clinical diseases. Much attention has been paid to the protective effects of natural antioxidants against drug-induced toxicities especially whenever free radical generation is involved [6, 7]. Popularity of herbal remedies is increasing and at least one-quarter of patients with liver disease use botanicals. The World Health Organization (WHO) estimates that 80 percent of the population of some Asian and African countries presently use herbal medicine for some aspects of primary health care. Some medicinal herbs have proven hepatoprotective potential [8, 9].
Medicinal plants have long been recognized as important sources of therapeutically active compounds [10]. Evidence-based research supports the medical and pharmacological benefits of plant-derived compounds with interest in the identification and characterization of their bioactive compounds [11 – 13]. Dialium guineense is a medicinal plant used in Traditional Medicine for the treatment of different disease conditions, such as diarrhea, severe cough, bronchitis, wound, stomachaches, malaria, jaundice, ulcer and hemorrhoids [14 – 16]. Although the acute toxicity of its extracts have been reported, little or nothing is known about the subchronic toxicity of the plant extracts [17]. This study investigated the subchronic toxicity of graded doses of extracts of D. guineense stem bark in rats.
MATERIALS AND METHODS
Chemicals and Reagents
All reagents used in this study were of analytical grade and they were bought from British Drug House (BDH) (England), Merck (Germany) and Sigma-Aldrich Ltd. (USA).
Plant Material and Authentication
Fresh stem barks of D. guineense were obtained from Auchi, Edo State, Nigeria and authenticated at the herbarium of the Department of Plant Biology and Biotechnology, University of Benin, Benin City, Nigeria (No. UBHD330).
Plant Extraction
Extraction of the pulverized plant material was by maceration over a 72 h period [18]. A portion (500 g) of the powdered stem bark was soaked in 5000 mL distilled water or absolute ethanol. The resultant aqueous and ethanol extracts were filtered with a muslin cloth and freeze dried using a lyophilizer.
Experimental Rats
A total of 70 adult male Wistar rats (35 per extract), which weighed between 160 and 180 g (mean weight = 170 ± 10 g) were purchased from the Department of Anatomy, University of Benin, Benin City, Nigeria. The rats were housed in metal cages under standard laboratory conditions: room temperature, 55 – 65 % humidity and 12-h light/12-h dark cycle. They were allowed free access to pelletized growers mash and clean drinking water. Prior to commencement of the study, the rats were acclimatized to the laboratory environment for seven days. Standard experimental protocol was followed for this study.
Subchronic Toxicity Study
The rats were divided into 7 groups (5 rats per group): Group I served as control, while rats in groups II – VII received graded doses of aqueous or ethanol extract (200 – 5000 mg/kg bwt) for a duration of 28 days. At the end of the 28th day the rats were fasted overnight and euthanized. The liver of all experimental rats were harvested, washed in ice–cold saline, blotted dry and placed in plain containers. Weighted portions of the organ were used to prepare 20 % tissue homogenate. The tissue homogenate was subsequently centrifuged at 2000 rpm for 10 min to obtain supernatant, which was used for MDA determination.
Determination of Lipid Peroxidation in Liver
Malondialdehyde (MDA) level was measured in liver homogenate [19].
Histological Examination of the Tissues
Sizeable portions of the liver were sectioned and fixed in 10 % formalin for 48 h, and thereafter dehydrated using varied concentrations of ethanol. Just before embedment in paraffin, the specimens were cleared thrice with xylene. Serial sections of 4 μm thickness were cut and stained with haematoxylin and eosin (H & E) according to standard protocol. Histopathological examination was carried out under light microscopy. In each H and E section, exactly 25 circular tubules were measured in two axes drawn perpendicular to each other with the aid of an image analyzer (Image Proplus, version 3.0).
Statistical Analysis
Count data are expressed as mean ± standard error of mean (n = 5). The statistical analysis was performed using SPSS (version 20). The various treatment groups were compared using Duncan multiple range test. Statistical significance was assumed at p < 0.05.
RESULTS
Effect of Graded Doses of Extracts of D. guineense Stem Bark on Rat Weight
As shown in Table 1, percentage increases in body weights of rats treated with aqueous or ethanol extract of D. guineense stem bark were significantly reduced, when compared with control group (p < 0.05). However, there were no significant differences in the corresponding relative organ weights among the groups (p > 0.05).
Table 1: Percentage Body Weight Increase and Relative Liver Weight of Rats Treated with Extracts of D. guineense Stem Bark
| Groups | % Increase in weight
Aqueous |
Relative organ weight (x 10-2) | % Increase in weight
Ethanol |
Relative organ weight (x 10-2)
|
| Control | 61.35 ± 4.11 | 3.80 ± 0.30 | 61.35 ± 4.11 | 3.80 ± 0.30 |
| 200 mg/kg bwt | 49.09 ± 4.83a | 4.10 ± 0.30 | 52.60 ± 2.92a | 3.00 ± 0.02 |
| 500 mg/kg bwt | 47.39 ± 3.09a | 3.60 ± 0.04 | 22.63 ± 1.56b | 4.70 ± 0.90 |
| 1000 mg/kg bwt | 42.38 ± 2.61a | 3.50 ± 0.05 | 21.00 ± 1.00b | 3.00 ± 0.02 |
| 2000 mg/kg bwt | 37.28 ± 3.94a | 4.10 ± 0.40 | 18.30 ± 1.06b | 3.40 ± 0.30 |
| 3500 mg/kg bwt | 31.65 ± 2.83b | 3.40 ± 0.10 | 17.73 ± 0.92b | 3.90 ± 0.60 |
| 5000 mg/kg bwt | 27.82 ± 0.40b | 4.00 ± 0.30 | 16.80 ± 1.10b | 4.10 ± 0.30 |
Data are percentage weight increase and relative liver weight, and are expressed as mean ± SEM (n = 3). ap < 0.05, when compared with control group; bp < 0.05, when compared with control and the other groups.
Effect of Graded Doses of Extracts of D. guineense Stem Bark on MDA Level
The concentrations of MDA in the liver homogenates of rats treated with graded doses of aqueous or ethanol extract were not significantly increased when compared with those of control group (p > 0.05; Table 2).
Table 2: Concentrations of MDA in Liver Homogenates
| Groups MDA Concentration (mole/mg protein) x 10-6
Aqueous Ethanol |
||
| Control | 6.99 ± 0.83 | 6.99 ± 0.83 |
| 200 mg/kg bwt | 6.92 ± 0.86 | 9.03 ± 2.72 |
| 500 mg/kg bwt | 7.32 ± 1.04 | 9.11 ± 2.74 |
| 1000 mg/kg bwt | 7.95 ± 1.15 | 9.61 ± 2.91 |
| 2000 mg/kg bwt | 10.57 ± 1.91 | 9.43 ± 2.51 |
| 3500 mg/kg bwt | 14.03 ± 3.12 | 12.08 ± 2.66 |
| 5000 mg/kg bwt | 14.56 ± 2.04 | 13.20 ± 2.48 |
Data are concentrations of liver MDA and are expressed as mean ± SEM (n = 5).
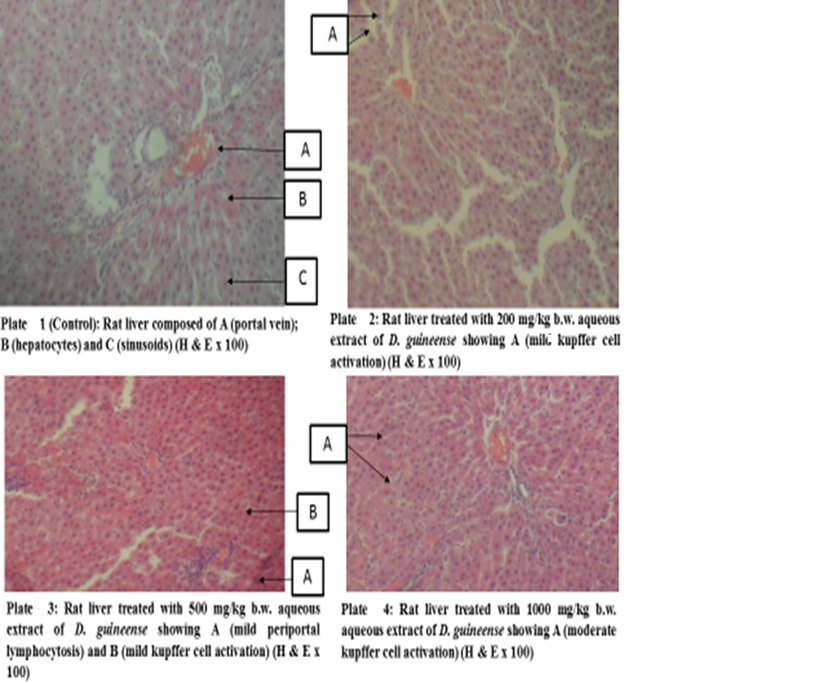
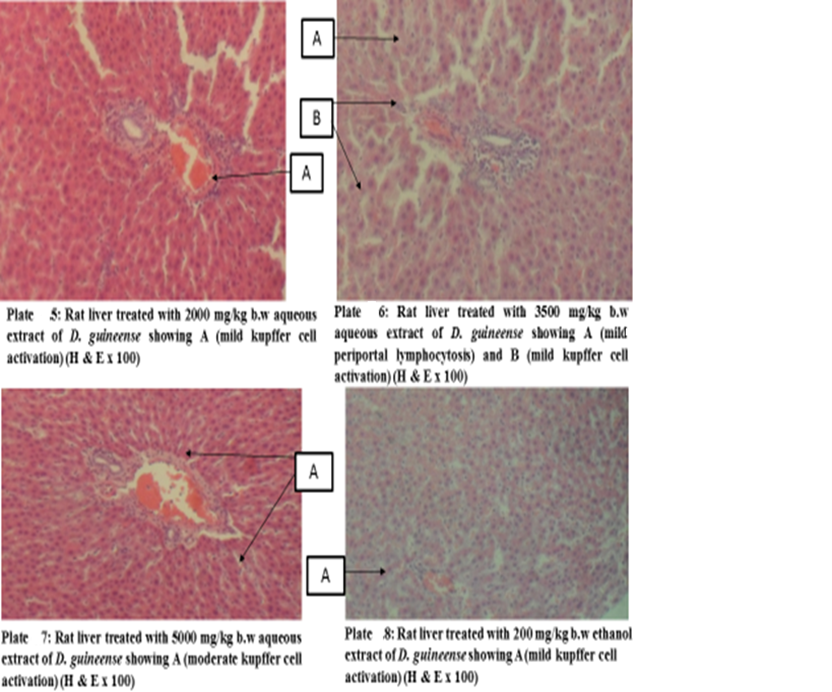
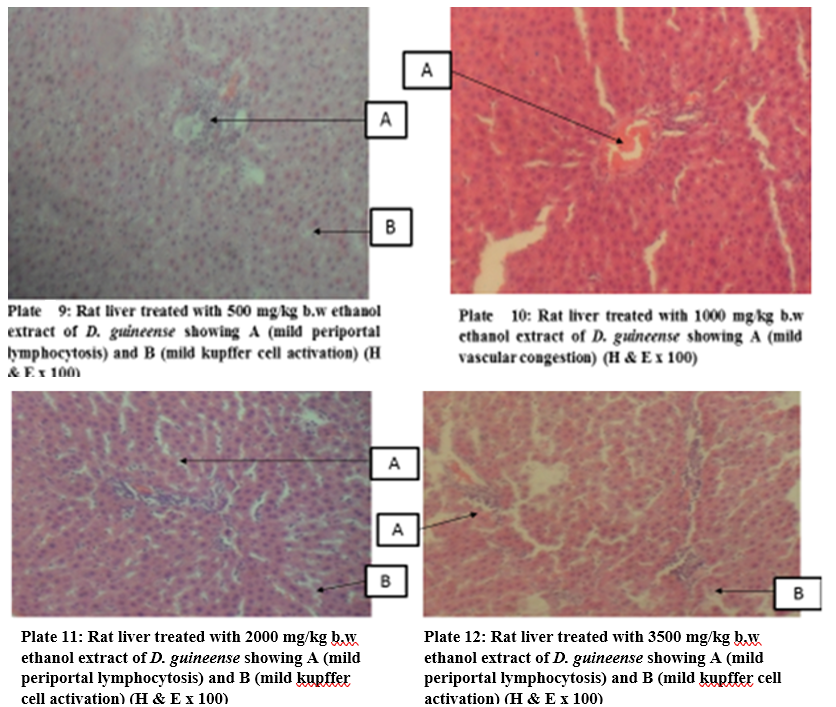
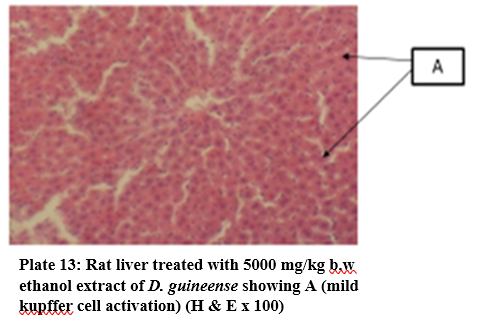
Figure 1: Effect of Graded Doses of Extracts of D. guineense Stem Bark on Rat Liver Histology
DISCUSSION
A major setback limiting the use of plant extracts in modern medicine is the inability to ascertain safe dose. Many medicinal plants known to be effective against a number of diseases have also been shown to be toxic at certain doses with prolonged exposure [17]. Drugs continue to be taken off the market due to possible hepatotoxicity.
As a result of its unique metabolism and close relationship with the gastrointestinal tract, the liver is susceptible to injury from drugs and other substances. Seventy five percent of blood coming to the liver arrives directly from gastrointestinal organs and then spleen via portal veins that bring drugs and xenobiotics in near-undiluted form. Several mechanisms are responsible for either inducing hepatic injury or worsening the damage process. The human body identifies almost all drugs as foreign substances (that is, xenobiotics) and subjects them to various chemical processes (metabolism) to make them suitable for elimination. This involves chemical transformations to reduce fat solubility and to change biological activity. Although almost all tissues in the body have some ability to metabolize chemicals, smooth endoplasmic reticulum in the liver is the principal “metabolic clearing house” for both endogenous chemicals (for example, cholesterol, steroid hormones, fatty acids and proteins) and exogenous substances such as drugs and alcohol [20]. Many chemicals damage mitochondria, an intracellular organelle that produces energy. Its dysfunction releases excessive amount of oxidants that, in turn, injure hepatic cells [21]. Activation of some enzymes in the cytochrome P-450 system such as CYP2E1 also leads to oxidative stress, which in turn has a crucial role in liver damage. Injury to hepatocyte and bile duct cells leads to accumulation of bile acid inside the liver. This promotes further liver damage [22, 23].
Changes in body weight serve as a sensitive indicator of the general health status of animals. Weight loss often synonymous with loss of appetite is due to disturbances in carbohydrate, protein or fat metabolisms [24].
Histology, also known as microscopic anatomy or microanatomy, is the branch of Biology which studies the microscopic anatomy of biological tissues. It is the microscopic version of gross anatomy which looks at larger structures visible without a microscope. In Medicine, histopathology is the branch of histology that includes the microscopic identification and study of diseased tissues [25 – 27]. In liver disease, several reports have documented gross and microscopic alterations of the morphology of hepatocytes [28, 29]. This study investigated the subchronic toxicity of graded doses of extracts of D. guineense stem bark in rats. The results showed that percentage increases in body weights of rats treated with aqueous or ethanol extract of D. guineense stem bark were significantly reduced, when compared with control group, but there were no significant differences in the corresponding relative organ weights and MDA levels among the groups. Similarly, the extracts did not significantly alter the normal architecture of the liver, but induced mild periportal lymphocytosis and kupffer cell activation. These results indicate that aqueous and ethanol extracts of D. guineense stem bark may be relatively safe, and agree with results of previous studies [30 – 58]. In addition, the extracts are likely to possess immunomodulatory activity.
CONCLUSION
The results of this study suggest that the extracts of D. guineense stem bark may not be deleterious to the liver and could contain agents that are potent activators of rat immune system. This study has provided a first-time evidence as to the relative safety of the plant extracts.
REFERENCES
- Gupta, S., Lonsdale, D. and Wang, D.P. (1994). The recruitment of estuarine copepod: A biological-physical model. Journal of Marine Research. 52: 687 – 710.
- Cotran, R.S., Kumar, V., Fausto, N., Nelso, F., Robbins, S.L. and Abbas, A.K. (2005). Robbins and Cotran pathologic basis of disease (7th ed.). St. Louis, MO: Elsevier Saunders. Pp. 878.
- Adi, A.H. and Alturkmani, H.J. (2013). Physiologically lucky: the role of medical physiology in modern medical education: an interview. Perspect Med Educ. 2: 99 – 103.
- Abu, O.D., Orobator, O.N. and Momodu, I.B. (2022a). Investigation of the Hepatoprotective Effect of Extracts of Dialium guineense Stem Bark in Wistar Rats Exposed to CCl Journal of Clinical Gastroenterology and Hepatology. 4 (2):123 -126.
- Abu, O.D., Orobator, O.N. and Momodu, I.B. (2022b). Evaluation of the Effect of Total Saponins and Tannins Isolated from Dialium guineense Stem Bark on CCl4 – Induced Hepatotoxicity in Wistar Rats. Global Journal of Medical and Clinical Case Reports. 9 (3): 035-038.
- Rai, M.K. (1994). Herbal medicines in India; retrospect and prospect. Fitoterapia. 65: 483 – 491.
- Stickel, F. and Schuppan, D. (2007). Herbal medicine in the treatment of liver diseases. Dig Liver Dis. 39: 293 – 304.
- Luper, S.A. (1998). Review of plants used in the treatment of liver disease: part one. Altern Med Rev. 3: 410 – 421.
- Thyagarajan, S.P., Jayaram, S., Gopalakrishnan, V., Hari, R., Jeyakumar, P. and Sripathi, M.S. (2002). Herbal medicines for liver diseases in India. J Gastroenterol Hepatol. 17: S370 – 376.
- Superling, F. (1979). Introduction to Toxicity Evaluation Session. Environmental Health Perspective. 32: 259.
- Walum, E. (1998). Acute Oral Toxicity. Environmental Health Perspectives. 106 (2): 497 – 503.
- Handa, S.S, Khanuja, S.P.S., Longo, G. and Rakesh, D.D. (2008). Extraction technologies for medicinal and aromatic plants. International Centre for Science and High Technology, Trieste. Italy. Pp. 21 – 25.
- Sandoval, M., Okuhama, N.N, Zhang, X.J, Condezo, L.A, Lao, J. and Angeles, F.M. (2002). Anti-inflammation and antioxidant activities of cat’s claw (Uncaria tomentos and Uncaria guinensis) are independent of their alkaloid content. Phytomedicine. 9: 325 – 337.
- Dalziel, J.M. and Hutchison, J. (1973). Flora of West Tropical Africa. Vol.1 (2nd Ed). The White friars Press Ltd. London. Pp. 561.
- Bero, J., Ganfon, H., Jonville, M.C., Frederich, M., Gbaguidi, F., De, M.P., Moudachirou, M. and Quetin, L.J. (2009). In vitro antiplasmodial activity of plants used in Benin in traditional medicine to treat malaria. Journal of Ethnopharmacology. 122 (3): 439 – 444.
- Arogba, S.S., Ajiboro, A. and Odukwe, I. (2006). A physicochemical study of Nigerianvelvet tamarind (Dialium guineense ) fruit. Journal of the Science of Food and Agriculture. 66 (4): 533 – 534.
- Abu, O.D. and Onoagbe, I.O. (2021). Acute toxicity of aqueous and ethanol extracts of Dialium guineense stem bark. Journal of Bioinnovation. 10 (2): 427 – 432.
- Abu, O.D., Imafidon, K.E. and Iribhogbe, M.E. (2015). Biochemical effect of aqueous leaf extract of Icacina trichanta on urea, creatinine and kidney oxidative status in CCl4– induced Wistar rats. Nigerian Journal of Life Sciences. 5 (1): 85 – 89.
- Guttridge, J.M.C. and Wilkins, C. (1982). Cancer dependent hydroxyl radical damage to ascorbic Formation of thiobarbituric acid reactive product. FEBS Lett. 137: 327 – 340.
- Blumenthal, M., Ferrier, G.K.L. and Cavaliere, C. (2006). Total sales of herbal supplements in United States show steady growth. Herbal Gram. 71: 64 – 66.
- Hozayen, W.G., Abou-Seif, H.S. and Amin, S. (2014). Protective effects of rutin and/or hesperidin against doxorubicin-induced hepatotoxicity. Int J Clin Nutr. 2 (1): 11 – 17.
- James, L.P., Mayeux, P.R. and Hinson, J.A. (2003). Acetaminophen-induced hepatotoxicity. Drug Metab Dispos. 31: 1499 – 1506.
- Jaeschke, H., Gores, G.J., Arthur, I.C., Jack, A.H., Pessayre, D. and Lemasters, J.J. (2002). FORUM: mechanisms of hepatotoxicity. Toxicol Sci. 65: 166 – 176.
- Eaton, D.L., Klaassen, C.D. (2001). Principle of Toxicology, in Klassen CD (ed) Casaret and Doll’s Toxicology. The Basic Science of Poisons. 6th McGraw-Hill. Pp. 11 – 13.
- Rosai, J. (2007). “Why microscopy will remain a cornerstone of surgical pathology”. Lab Invest.87 (5): 403 – 408.
- Titford, M. and Bowman, B. (2012). “What May the Future Hold for Histotechnologists?”. Laboratory Medicine. 43(2): e5 – e10.
- Wick, M.R. (2019). “The hematoxylin and eosin stain in anatomic pathology – An often-neglected focus of quality assurance in the laboratory”. Seminars in Diagnostic Pathology.36 (5): 303 – 311.
- Abu, O.D., Aleogho, B.M. and Omoregie O. (2019). Aqueous leaf extract of Icacina trichanta Oliv. improves lipid profile and CCl4 – induced histological changes in the liver and kidney of Wistar rats. Asian Journal of Research in Biochemistry. 4 (1): 1 – 11.
- Abu, O.D., Olude, O.M. and Obayuwana, H.O. (2021). Effect of methanol extract of Citrullus lanatus seed on hematological profile and tissue histology of normal Wistar rats. Advance Research Journal of Medical and Clinical Science 7 (7): 608 – 615.
- Abu, O.D., Onoagbe, I.O., and Ekugum E. (2022a). Hepatotoxicity of Graded Doses of Ethanol Extract of Dialium guineense Stem Bark in Wistar Rats. Journal of Pharmaceutical and Bio-Medical Sciences. 2 (9): 347 – 352.
- Abu, O.D., Onoagbe, I.O., and Ekugum E. (2022b). Nephrotoxic Evaluation of Aqueous Stem Bark Extract of Dialium guineense in Normal Wistar Rats. Journal of Pharmaceutical and Bio-Medical Sciences. 2 (9): 353 – 357.
- Abu O.D., Alegun O. and Ojo A.U. (2022). Pancreatotoxicity of Ethanol Extract of Dialium guineense Stem Bark in Rats. World Journal of Pharmaceutical and Life 8 (11): 40 – 45.
- Abu O.D., Okuo A.V. and Ayele P.E. (2022). Pancreatotoxic Effect of Aqueous Extract of Dialium guineense Stem Bark in Wistar Rats. International Journal of Novel Research in Life Sciences. 9 (5): 31 – 37.
- Abu, O.D., Odagwe B. and Ojo A.U. (2022). Cardiotoxicity of Ethanol Extract of Dialium guineense Stem Bark in Rats. World Journal of Pharmaceutical and Life Sciences. 8 (11): 34 – 39.
- Abu, O.D., Umar A-B. and Adekanle E. (2022). Cardiotoxic Effect of Aqueous Extract of Dialium guineense Stem Bark in Wistar Rats. East African Scholars Journal of Agriculture and Life Sciences. 5 (9): 167 – 172.
- Abu O.D., Iyare E. and Omoruyi I.J. (2022). Toxic Responses of the Blood of Rats Exposed to Aqueous Extract of Dialium guineense Stem Bark. FUDMA Journal of Science. 7 (2): 117 – 120.
- Abu O.D., Avenbuan E. and Ayele P. (2022). Investigation of the haematotoxicity of ethanol stem bark extract of Dialium guineense in rats. Journal of Clinical and Biomedical Advance, 2(1): 1-4.
- Abu, O.D., Ngedaa, OS and Osarhenomase, E.G. (2022). Effect of Extracts of Dialium guineense Stem Bark on Lipid peroxidation Index and Histological Changes in Kidneys of Normal Rats. Forensic Medicine. 4 (2): 23 – 29.
- Iyorah, I., Abu, O. and Ilegar, E. (2022). Oxidative stress responses to heavy metal burden in African Catfish, Clarias gariepinus from Warri River, Niger Delta, Southern Nigeria. Nigerian Journal of Veterinary Medicine. 21(2): 74-82
- Abu, O.D., Onoagbe, I.O., and Ohikhuare, F. (2022). Nephrotoxic Evaluation of Ethanol Stem Bark Extract of Dialium guineense in Normal Wistar Rats. International Journal of Forensic Medicine. 4 (2): 19 – 22.
- Abu O.D., Ezike T.V. and Ajuwa O.I. (2022). Cardioprotective property of extracts of Dialium guineense stem bark in rats exposed to CCl4. American Journal of Biomedical Science and Research. 2022: 689 – 693.
- Abu O.D., Umar A-B. and Ajuwa O.I. (2022). Protective Property of Total Saponins and Tannins of Dialium guineense Stem Bark in CCl4-Induced Cardiotoxicity in Rats. World Journal of Genetics and Molecular Biology. 1 (1): 1 – 6.
- Abu O.D., Iyare E. and Ogboi K.U. (2022). Cardiac Oxidative Status in CCl4-Exposed Rats Treated with Extracts of Dialium guineense Stem Bark. Global Journal of Scientific Frontier Research. 22 (01): 1 – 6.
- Abu O.D., Iyare E. and Ogboi K.U. (2022). Antioxidant Property of Total Saponins and Tannins of Dialium guineense Stem Bark in Rats Hearts Exposed to CCl4. Journal of Clinical Epidemiology and Toxicology. 3 (3): 1 – 4.
- Abu O.D., Umar A-B. and Eiremiokhae O. (2022). Investigation of the Cardioprotective Capacity of Aqueous Extract of Icacina trichanta Leaves in Rats Exposed to CCl4. Journal of Genetics and Cell Biology. 6 (1): 322 – 328.
- Abu O.D., Eromosele, A.I. and Osarhenomase, E.G. (2022). Effect of Extracts of Dialium guineense Stem Bark on Lipid Profile and CCl4– Induced Histological Changes in Liver of Wistar Rats. International Journal of Lipids. 1 (1): 22 – 27.
- Abu O.D., Omage J.I. and Ogbebor E.O. (2022). Effect of Total Saponins and Tannins Isolated from the Stem Bark of Dialium guineense on Lipid Profile and CCl4– Induced Histological Changes in Liver of Wistar Rats. Journal of Medicine and Biology. 3 (2): 1 – 9.
- Abu, O.D., Awhin, E.P. and Ohikhuare, F. (2023). Effect of Methanol Fraction of Ethanol Extract of Dialium guineense Stem Bark on Cardiovascular Disease Risk Factors in Diabetic Rats. Journal of Biology and Medicine, 4 (1): 128.
- Abu, O.D., Awhin, E.P. and Iyare, H.E. (2023). Investigation of Renal Function in Diabetic Rats Treated with Methanol Fraction of Ethanol Extract of Dialium guineense (MEDG) Stem Bark. Journal of Urology and Nephrology Studies, 4 (4): 513 – 518.
- Abu, O.D., Obaze, G.E., Egili, S. and Idehen, I.O. (2023). Ethanol Extract of sativus Modulates the Activity of Glucose 6-phosphatase/ Aminotransferases and Levels of Lipids in Tissues of STZ-Induced Diabetic Rats. Biomedical Journal of Scientific and Technical Research. 53(4): 44989-44994.
- Abu, O.D., Ojo, I. and Awhin, E.P. (2023). Protective Property of Ethanol Extract of sativus on STZ-Induced Diabetic Rat Pancreas. Biomedical Journal of Scientific and Technical Research. 52(2): 43613-43618.
- Abu, O.D., Ojo, I. and Ezike, T.V. (2023). Methanol Fraction of Ethanol Extract of Dialium guineense Stem Bark Mitigates STZ-Induced Oxidative Stress in Rat Biomedical Journal of Scientific and Technical Research. 51 (2): 42594 – 42600.
- Abu, O.D., Osime, E.C. and Ngedaa, O.S. (2023). Cardiac Oxidative Status in Diabetic Wistar Rats Exposed to Ethanol Extract of Cucumis sativus J. Diagnostics and Case Reports. 4 (2): 1 – 5.
- Abu O.D., Awhin E.P. and Ozedu M.E. (2023). Evaluation of Cardiovascular Disease Risk Factors in Diabetic Rats Administered Ethanol Extract of Cucumis sativus Fruit. African Journal of Health, Safety and Environment. 4(1): 108 – 117.
- Abu O.D., Awhin E.P. and Iyare H.E. (2023). Assessment of Renal Function in Diabetic Wistar Rats Treated with Ethanol Extract of Cucumis sativus. African Journal of Health, Safety and Environment. 4(1): 101-107.
- Abu O.D., Awhin E.P. and Ifekwe, J.C. (2023). Liver Function Status of Diabetic Wistar Rats Treated with Ethanol Extract of Cucumis sativus Biomedical Journal of Scientific and Technical Research. 51 (2): 42440 – 42445.
- Abu O.D., Ohikhuare F. and Ezike T.V. (2023). in vitro Antioxidant Activity of Aqueous and Ethanol Extracts of Cucumis sativus. Journal of Clinical Epidemiology and Public Health. 01 (03): 1 – 6.
- Abu, O.D., Alegun, O. and Ifekwe, J.C. (2023). Renal Oxidative Status in Diabetic Wistar Rats Administered Methanol Fraction of Ethanol Extract of Dialium guineense. Medical and Clinical Case Reports Journal. 1(1): 1 – 13.